Camille Alphonse Faure was a French chemical engineer who improved the performance of early lead acid batteries. His compatriot, Gaston Planté’s breakthrough version may have been a great step forward. But it needed Camille Faure’s coated lead sheet solution to make it truly viable. Read more to discover the science and history behind this development.
Story Behind Camille Faure’s Coated Lead Sheet Idea
The lead acid battery was an overnight sensation that literally had telegraph wires buzzing. That’s because it delivered sufficient energy to make the arc lamps at the 1878 Paris Exposition seem almost a side show.
But it needed the extra oomph of Camille Faure’s intervention to propel the electric cars already forming in inventive minds. Drivers would need better driving ranges, and higher top speeds if the trend was to take off.
Fauré’s stroke of genius involved coating the battery lead sheets with a paste he made from lead oxides, sulfuric acid and water. He then contrived a way to cure the coating in a warm, humid atmosphere. This then became the chemically active material, when he charged the battery and made it good to go.
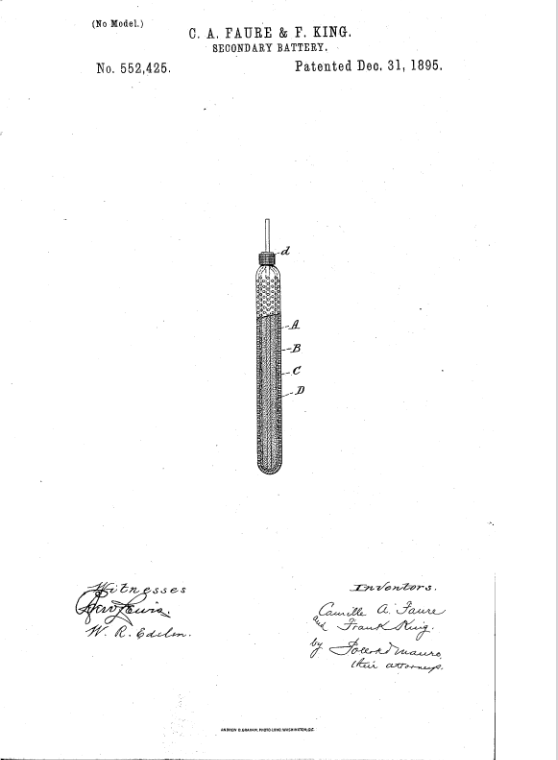
The Technology Behind The Illustration
Camille Faurés patent explains how he covered the lead plates with a “thin sheet, or layer of silicate asbestos fabric, or cloth”. This material was porous and could not oxidize. So he placed it in a “perforated envelope of celluloid, ebonite, or other sufficiently acid-resisting non-conducting material.”
Our image above shows a cross-section of the result. The patent explains how “A is the conducting support, while B is the active material. C is the non-conducting porous sheet of silicate asbestos. Finally, D is the “perforated envelope or cover of celluloid or ebonite, which C prevents from contacting the peroxide of lead of the electrode”.
More Information
Planté’s Choice of Lead & Why He Made It




