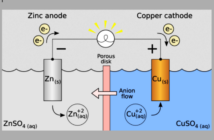
Planté’s choice of lead was an unexpected departure from a convention of using copper and zinc. We decided to explore how the Frenchman made his decision in 1859, and so became the father of lead-acid batteries. Strangely, we did not find much on Google on the topic. Although we did already know the pure metal is a good conductor due to the presence of free electrons.
Did Free Electrons Influence Planté’s Choice of Lead
Free electrons are not attached to an atom or a molecule, and are therefore free to respond to an external source of energy. And this phenomenon allows them to conduct an electric current too. Wikipedia explains how Planté explored the differences between static and dynamic electricity, and this may have involved experimenting with different metals.
So we can imagine him picking up a piece of lead, and wondering whether the ‘solid lump’ might conduct more electricity than a thin sheet of zinc or copper. Perhaps Planté noticed how pure lead oxidized in the air, and this prevented the flow. After all, those intrepid scientists were ‘shooting half blind’ in a world of science they did not fully know.
Planté Assembles the First Rechargeable Lead Battery
Our man of the moment took a leap of faith on a day in 1859. First he took two sheets of lead, and rolled them in a spiral with a linen cloth between them. And then he placed the roll in a glass jar of sulfuric acid solution, and produced electricity! This was a wonderful breakthrough, especially when he discovered he could recharge it with an external source.
Linda Hall Library explains how Planté assembled 20 separate cells into the 1860 battery at Science Museum in our image. Apparently ‘an elaborate switch at the top allowed the cells to be switched from parallel to series’. Lead batteries were finally making progress, with many familiar elements already in place.
More Information
Cruickshank Trough Eliminates a Pile of Woes