We spoke about electrode potential in a previous post, and the need to balance it so the electrons travel effectively. Today we turn our attention to how overall battery potential determines voltage. And voltage, as we know is the pressure propelling charged electrons through a circuit.
Optimizing Battery Potential to Increase Voltage
The differential in the standard potential of the electrodes determines the force with which electrons travel, and hence the voltage. Should a researcher wish to increase that force, then they would have two options:
- Choose different electrode materials to increase electrochemical potential.
- Stack several cells in series to multiply the force with which the electrons travel.
However, should the researcher connect several cells in series instead, then the voltage would not increase. But the potential current should, being the total number of electrons flowing through the battery cells.
The Role of the Electrolyte in This Chemistry
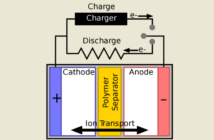
We now understand how overall battery potential determines voltage, and how we could increase this. Now we turn our attention to the electrolyte. What does it do, and how does this influence battery performance. Do you remember how the first battery had paper soaked in saltwater separating the discs?
Nowadays we have many more electrolytes to choose from, of course. They can be liquid, gel, or solid. However, they must all allow charged ions to pass through.
Remember this very important point. Electrons hold a negative charge. Therefore, we need a way to balance their negative movement through the electrical circuit. This is where the electrolyte makes the difference. It is the medium through which the balancing, positive ions flow.
More Information
The Chemistry of an Electric Battery