Rechargeable zinc-bromine batteries are not battery industry whizz kids, aiming to knock lithium-ion off its pedestal. No, not at all, in fact they just want to be solid citizens doing a decent job. This makes them ideal for grid storage, and stand-alone power systems. They also happen to be relatively inexpensive and less likely to catch fire too.
What Happens Inside Zinc-Bromine Batteries?
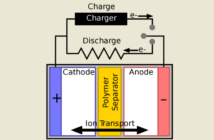
All batteries use an electrochemical chemical process to store and deliver electricity. Well, electrical energy actually, but it boils down to the same thing. Our batteries of interest in this post use a chemical reaction involving zinc metal and bromine, hence their particular name.
The electrolyte that controls the process is an aqueous solution of zinc-bromide. All the materials are readily available, and relatively inexpensive and stable. And as a result these batteries are Iess likely to overheat, and potentially catch fire than lithium-ion ones.
Comparing Them Again With Lithium-Ion Batteries
Rechargeable zinc-bromine batteries score better than lithium-ion ones in the following aspects:
- They readily tolerate a 100% depth of discharge on a daily basis.
- Their capacity decreases gradually over more than 5,000 cycles.
- Their electrolyte is nonflammable and hence they are a low fire risk.
- They do not heat up, and therefore do not need cooling systems.
- Their materials are low-cost and readily available on the market.
- Their design makes it easy to dismantle and recycle their materials
That said these batteries are not the perfect solution for every application. They do not compete well with lithium-ion across these dimensions:
- They have relatively low energy density, producing a lower voltage.
- They also have to be fully discharged at least every couple of days.
These last two factors make zinc-bromine batteries unsuitable for phones, laptops, and other mobile applications. Although they are ideal for supporting daily-cycling operations such as solar generation and grid support, and are available in both cellular and flow versions.
More Information
Key Battery Materials Deep in the Ocean