A team at Harvard’s John A. Paulson School of Engineering and Applied Sciences claims a new lithium metal chemistry. This appears to be a ground-breaking solution for lithium dendrites, and presents a model for a practical solid-state battery. Could this be the paradigm-shifting step our world of batteries has been waiting for? We paged through the Harvard Office of Technology Development announcement to learn more.
New Prototype Battery Resolves Lithium Dendrite Formation
The Harvard announcement describes how dendrites form on lithium-metal batteries during charging, as follows:
- Plating on the anode occurs when lithium ions arrive from the cathode.
- This creates “an uneven, non-homogeneous surface, like plaque on teeth”.
- The uneven plating strips selectively during discharging, creating “potholes”.
- The dendrites take root, and grow steadily during “repeat recharging”.
Associate Professor at John A. Paulson School, Xin Li, previously published a paper concerning a “multi-layer battery”. This “sandwiched different materials of varying stabilities between the anode and cathode”, and controlled lithium dendrite penetration without stopping it.
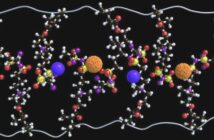
However, the new work in which Xin Li is senior author takes this a step further. That’s because it uses, “micron-sized silicon particles in the anode to constrict the lithiation reaction”. And this solution for lithium dendrites “facilitates homogeneous plating of a thick layer of lithium metal”.
Does this Bring Us Closer to a Lithium-Metal Pouch Battery?
The Harvard announcement explains how their design prevents the ions penetrating the anode deeply during recharging. They remain on the surface, unlike the deep penetration characteristic of lithium-ion batteries.
“In our design, lithium metal gets wrapped around the silicon particle”. This is, “like a hard chocolate shell around a hazelnut core in a chocolate truffle,” Xin Li explains.
Plating and stripping occurs rapidly on a uniform surface. The evenly-distributed current density allows fast plating and stripping. The battery can recharge in only about ten minutes, they say.
A prototype, stamp-sized pouch cell version retained 80% capacity after 6,000 cycles, way ahead of other options. Harvard Office of Design Technology has licensed a spin-off company, involving Xin Li, to build a smart-phone-sized pouch battery.
More Information
Lightweight Lithium Metal Takes Center Stage
3-D Exposes Dendrites Forming in Batteries