Surgeons and doctors use a topical iodine named betadine as first-aid for wounds, in order to prevent infection. Although in laboratories they call the preparation povidone-iodine, and prepare it by blending iodine with a polyvinylpyrrolidone polymer. But that’s enough for science. What on earth has this to do with ultra-fast charging batteries?
The Numerous Advantages of Lithium-Sulphur Batteries
Researchers at Monash University in Melbourne, Australia have developed ultra-fast charging batteries, using lithium-sulphur chemistry. They report that these batteries are potentially powerful enough to drive ‘long-haul electric vehicles and commercial drones’.
Lithium-sulfur batteries already have potential to replace the current range of lithium-ion cells. Here are some of the reasons why battery scientists are excited by this possibility:
- The low atomic weight of lithium, and moderate atomic weight of sulphur, make the batteries as light as water.
- Lithium-sulphur batteries deliver more energy than lithium-ion ones, because they have higher battery density.
- Lithium-sulphur batteries are also cheaper, because they do not use cobalt and / or iron compounds.

This promising chemistry has, however, not been generally adopted. This is because the active material steadily leaks from the cathode, curtailing the number of recharges. And, furthermore, the low conductivity of the sulphur cathode means it needs to be larger, expanding the overall size of the battery.
Ultra-Fast Charging Batteries With Betadine Inspiration
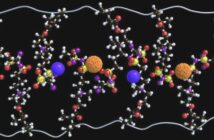
The team members believe that their inspiration came from the chemistry of betadine, which is a common household antiseptic. And that their idea translated into a way to accelerate the charge and discharge rates of lithium-sulfur batteries.
We turned to the research report that we link to below and established the following technical explanation:
- Polyiodide species held within a complex polar network of polyvinylpyrrolidone, accelerate the oxidation and reduction phases of battery recycling.
- The material achieves this by accelerating the reaction pathways mediated by inter-valance polyiodide reactions, within the working voltage of lithium-sulfur.
“Imagine an electric vehicle that can travel 625 miles from Melbourne to Sydney on a single charge,” says one researcher. “Or a smartphone that charges in minutes – we are on the cusp of making this a reality.”
More Information
Lithium-Sulfur Battery Capacity Innovation
Sulfur Batteries Soar with Wings
Preview Image: Campus Building at Monash University
Monash University News of November 28, 2024