An iron redox flow battery – also known as a salt battery – stores energy through an electrochemical reaction of iron. This chemistry could be a viable alternative to lithium-ion, on the basis of the low cost of the iron water and air battery components. These three materials also do not burn, making this technology especially attractive.
First Iron Water and Air Battery On a Grid
The size and weight of iron redox flow batteries has discouraged grid storage applications until now. However despite this, New Scientist advises that disruptive startup Ore Energy, appears to have broken through.
The Netherlands-based company has achieved a notable success, by storing 100 hours or more of energy in a iron redox flow battery. Moreover, it has successfully delivered the energy to a utility at Delft University of Technology.
What Iron Water and Air Mean For Renewables
Renewable battery storage needs more than a few hours of capacity, to be able to support a grid during a lengthy storm. Or else – unless we can achieve this – renewables could remain an auxiliary electricity source for ever.
Most grid-connected storage batteries until now, have been the lithium-iron phosphate variety. But these can only deliver electricity for four-to-six hours, which is not nearly good enough.
To add to the conundrum, lithium-iron phosphate materials are expensive, and some are flammable too. Whereas the iron water and air battery from Ore Energy, should have neither of these two potential deal-breakers.
Rusting is a Simple, Well-Known Chemical Reaction
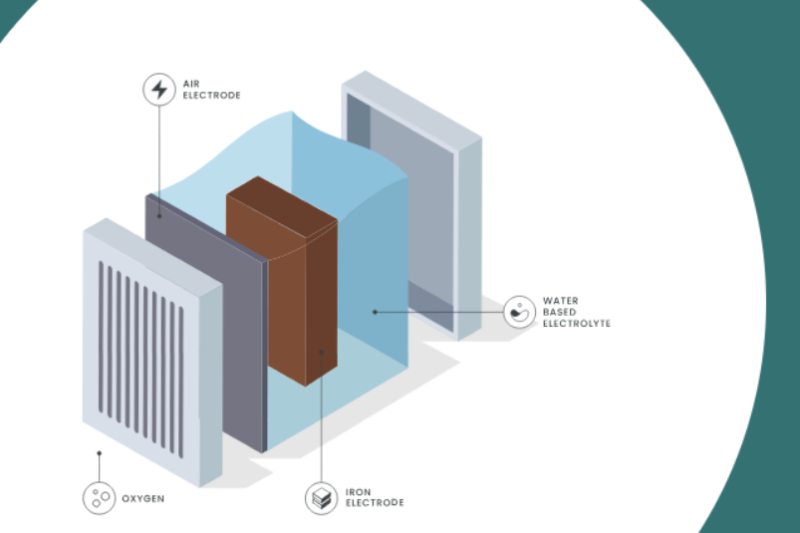
Rusting is a simple, well-known reaction, as Ore Energy explains on its website. First, the metallic iron oxidizes as rust while discharging, and gives off an electrical current of electrons.
And then, when it is time for the redox flow battery to recharge, an external electrical current reverses the rust, and reinstates the metallic iron. Could the solution be as simple as that?
More Information
Iron Flow Batteries Touch Down At Schiphol




