Coulombic and Faradaic efficiency both rate the ability of charge-carrying ions to transfer their energy correctly in electrochemical batteries. Although of course these terms have other applications beyond the scope of this article. Here we focus on coulombic efficiency in batteries as a measure of energy storage effectiveness, which is so very important nowadays.
Where Does the Term Columbic Efficiency Come From?
Our story begins back in the 18th Century, when English Chemist Michael Faraday was studying electromagnetic induction, diamagnetism, and electrolysis. He had little formal education, yet Faraday was to become one of the most influential scientists in history.
Here are two things that are particularly relevant to our world of batteries:
- Scientists originally referred to a unit of electrical charge as a ‘Faraday’, although nowadays we call it a ‘Coulomb’.
- The ‘Faraday Constant’ is the total electric charge, divided by the total amount of electrons involved.
Both these factors are essential inputs when we measure coulombic efficiency in batteries. We now turn to examining how electrochemical batteries lose some of their ‘Faraday Constant’ charge.
How ‘Faradaic Losses’ Occur in Electrochemical Batteries
‘Faradaic Losses’ can occur in both galvanic and electrochemical cells. Galvanic cells convert chemical energy into electrical energy, through a spontaneous redox reaction. Whereas electrochemical cells do the same thing during a controlled redox process, as do batteries too.
However, despite these controls, batteries still experience Faradaic Losses that shorten their useful operating life. There are three main reasons for this phenomenon, as follows:
- There could be a gassing reaction, such as hydrogen evolution at the negative electrode.
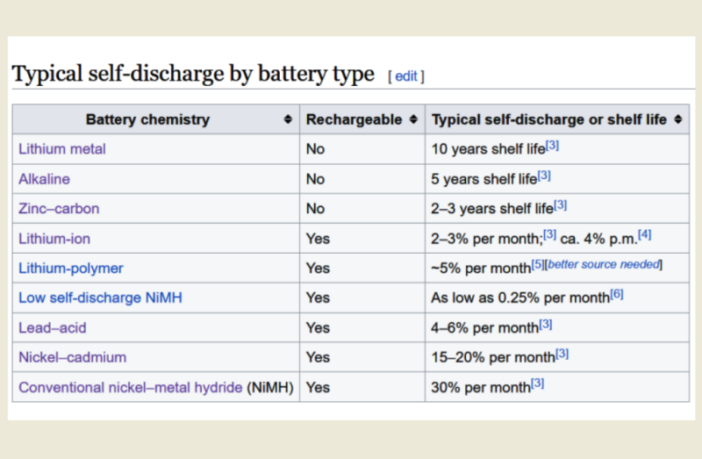
- Automatic self-discharging could occur through an unwanted chemical reaction consuming ions.
- Ions could flow directly between electrodes through internal pathways, bypassing an external circuit.
These losses reducing coulombic efficiency in batteries, can occur when a battery is in operation, or when a battery self-discharges in storage.
More Information
Birth of the Battery – Where We Came From
Battery Self Discharging in Focus at Argonne