Scientists at Limerick University in Ireland, co-developed a world-first dual lithium-sodium battery that could reshape the future of electricity storage. Their innovative full-cell, dual-cation battery combines lithium and sodium cations. This ‘significantly enhances’ battery stability and storage capacity, and could take us through new frontiers in battery science.
The Dual Power of Lithium-Sodium Batteries
This achievement is the result of collaboration between scientists at Limerick University in Ireland, and University of Birmingham in England. In a nut shell, their novel battery:
- Combines the strengths of sodium and lithium metals for better performance.
- Uses sodium as the main component for greater efficiency and sustainability.
“For the first time, we’ve shown that we can ‘supercharge’ sodium-ion batteries, by pairing sodium and lithium in a sodium-dominant dual-cation electrolyte,” a team member explains. “This breakthrough opens the door to more sustainable, high-performance battery chemistries.
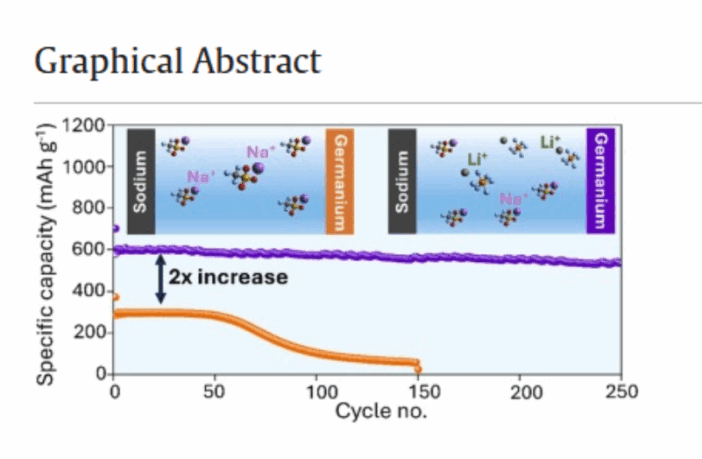
“By introducing both lithium and sodium cations, we actually double the battery’s capacity, that would otherwise be lower in a typical sodium-ion battery. Nobody has done this before with the anode materials we chose.”
Digging Down Into the Chemistry Behind the Battery
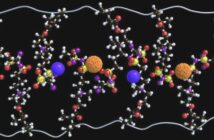
Sodium and lithium cations are positively charged ions, that have lost one or more electrons. This results in those atoms or molecules carrying a positive charge. They move between the anode and cathode as a battery discharges. Recharging a battery sends them back to the anode again.
Our world-first dual lithium-sodium battery allows lithium to become the ‘capacity booster’, the scientist continues. This ‘supercharges’ the sodium-ion system with additional storage density, while retaining sodium’s long-term stability.
Sodium-ion batteries have failed to make inroads into lithium-ion’s less-stable world, because consumers want batteries that last longer between recharges. This discovery opens a new door to sodium-ion chemistry, which is far less likely to overheat, catch fire, and potentially explode.
More Information
Natural Tunnels for Sodium Ions
Sodium-Ion Batteries Ringing The Bell
Preview Image: Without and With Lithium Cations