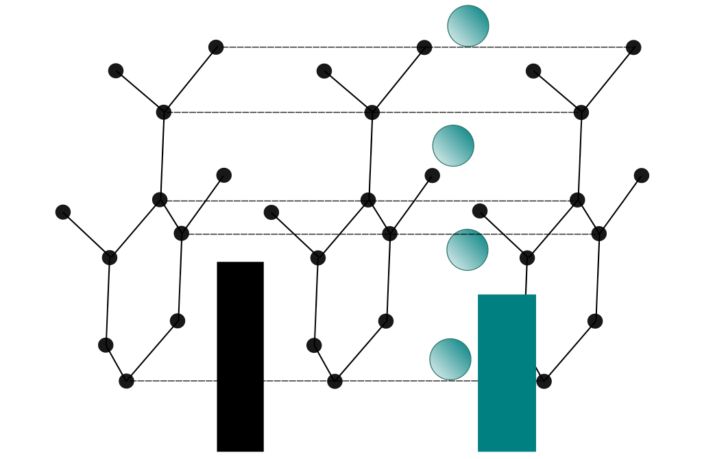Battery intercalation is a complex topic. However we dedicated this website to explaining battery basics. Thus we are going to keep this conversation at a high level. ‘Intercalation’ is the temporary insertion of something into something else. For example, every fourth year we intercalate an extra day into our calendar. Then we remove it again the following year.
Intercalation in Chemistry

In chemistry, intercalation is the reversible inclusion of a molecule, or an ion, into a compound with a layered structure. Stated simply, it slips in between the strata.
In this regard you may find it helpful to imagine something like these strata in New South Wales, Australia. In this instance the process is however not reversible.
That said, the commonest layered structure appearing naturally is graphite, although transition metal dichalcogenides are another source. Many of these are sulphides. In this context you may recognize titanium disulphide and cadmium diodide from your battery research. At this point, we are going to bail out. We want to return to our favorite topic which is batteries.
Getting Back to Battery Intercalation
Ions travel from the anode to the cathode when we discharge a battery. Battery intercalation occurs when the ions temporarily insert themselves between layers in the cathode. Then we can return them to the anode by recharging if it is a renewable battery.

The efficiency of battery intercalation is fundamental to battery aging. For example, there are capacity and density limitations to the materials we use for cathodes, including graphite .
Lithium battery research is ongoing concerning reversible chemical conversion as opposed to intercalation.
We never cease to wonder how batteries light up our lives. Every month scientists make groundbreaking new discoveries. Behind the scenes, battery intercalation continues to play its vital role. It follows that without it we might be unable to consume energy harvested from the wind and sun.
Related