When you switch on your smartphone or your smartwatch, you expect them to start working instantly. They generally do so if their battery holds a charge, thanks to the movement of cations and anions inside the battery. Most folk don’t realize that their batteries only work, because these tiny particles inside the battery are constantly on the move.
More About Moving Cations and Anions
If those movements stopped, then your smartphone or your smartwatch would not work. We call those particles ‘ions’, although they are actually atoms carrying electrical charges. Cations are atoms with positive charges. Anions have negative charges which makes the difference.
This difference matters because opposite charges attract, and like charges repel. This simple fact is behind the movement of cations and anions in batteries. It may help you to remember experiments with magnets when you were at school (although they use a different form of energy).
When a Battery Discharges
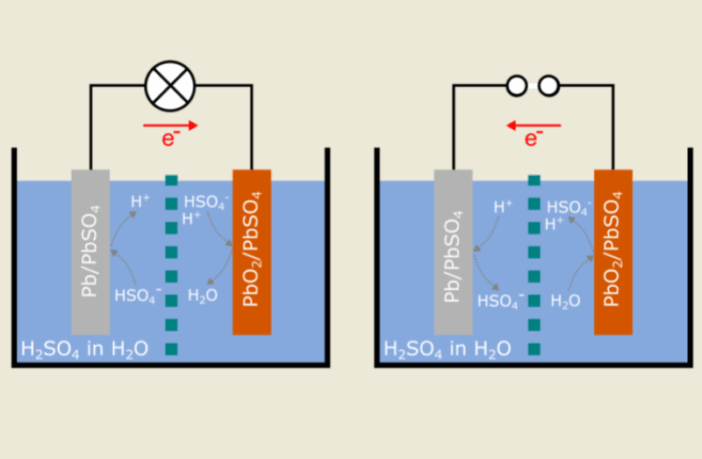
When a battery discharges, it sends electrons (think little bits of electricity) from its anode to its cathode via a circuit OUTSIDE. This creates an imbalance INSIDE the battery. To correct this imbalance, positive cations move from the cathode to the anode INSIDE the battery, while negative anions move in the opposite direction to the cathode INSIDE the battery.
When a Battery Recharges
When a battery recharges from an OUTSIDE source, the battery returns to its original state, minus a few cations and anions lost in the process. The charger sends a fresh supply of electrons to the anode. Meanwhile, the remaining cations and anions return to the cathode and anode respectively.
All Batteries Work the Same Way
All batteries use electrons, cations, and anions regardless of their type. Those principles apply to all types of batteries, including lithium-ion, lead-acid, and even basic alkaline cells.
Different batteries use different materials, but they all rely on the coordinated movement of cations and anions. Each and every one depends on the movement of cations and anions inside, to do their work they could not do without them.
More Information
Electrons Versus Ions in Batteries