Batteries are not like living things that breathe in oxygen to stay alive. Although some batteries do need oxygen to start working. All the other types are like packaged lunches, with everything on board. So let’s investigate the difference, and discuss how batteries that breathe oxygen work
The System That Help Batteries Breathe
Some batteries take in air, although this does not mean that they are alive. Most other batteries, including lead-acid and lithium-ion, are self-contained devices, with everything they need to work inside.
But air breathing batteries are different because one side is open to the air. They take this air in through a special filter. As the air enters, its oxygen reacts with the rest of the battery. This how batteries that breathe in oxygen work, and produce their electricity.
The air circulates in through the battery and out again. Not requiring space to store oxygen means the battery can be smaller, lighter, and more powerful for its size.
Main Battery Types That Use Oxygen
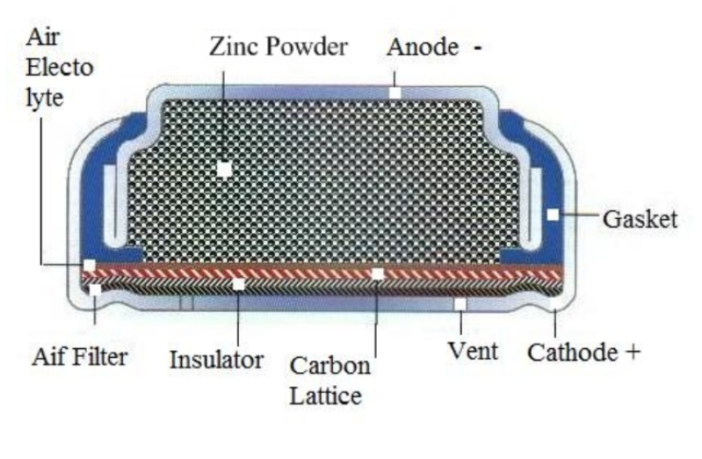
Zinc-air batteries typically produce 1.4 to 1.45 volts, and are the most popular air-breathing batteries. We find them in hearing aids and some medical devices.
Users start zinc-air batteries by removing stickers, so the air can enter and begin the process. This reaction continues until the battery is flat, or the user disconnects it from the device.
Zinc-air batteries are a special class of metal-air batteries that work similarly Other popular examples include aluminum-air and iron-air batteries. Lithium-air batteries are mostly still at the experimental stage.
Oxygen is an abundantly available material that supports life on our planet. We should cherish it, and use it responsibly, like all other battery resources.
More Information
Iron-Air Electro-Chemical Cells That Rust