The batteries we use in our homes store energy, which they deliver on request. Lithium-ion and lithium-metal batteries store this energy inside tiny atomic particles that chemists call ‘ions’. The lithium ions in household batteries have lost one electron, and so each ion carries a positive charge. This is the origin of the energy inside batteries.
Where Are These Lithium Ions in Our Batteries?
Almost all batteries, including lithium-ion and lithium-metal versions, have a pair of solid electrodes. One is the negative and the other is the positive. These connect to terminals that deliver electrical energy to the outside world.
Any electrolyte, which may be a liquid or solid material, keeps the electrodes physically apart. However, the electrolyte allows lithium ions to move between the electrodes. This is how a battery produces useful electrical energy.
The lithium ions in household batteries accumulate in different electrodes, depending on the state of charge, and whether the battery is discharging or charging:
- Lithium ions move from the negative electrode to the POSITIVE ELECTRODE as a lithium-ion battery releases its energy. This is where the electricity COMES FROM to power our phones, laptops, electric vehicles, and more.
- Lithium ions move from the positive electrode to the NEGATIVE ELECTRODE as we RECHARGE the lithium-ion battery’s energy. This is where the electricity GOES TO when we charge our batteries from an external source.
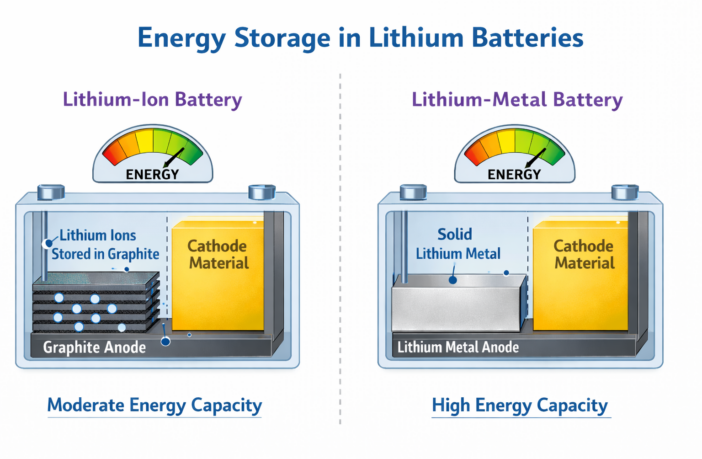
There’s a subtle difference with lithium ions in lithium-metal household batteries you should know about. These batteries are similar to lithium-ion versions except for one thing:
- LITHIUM-METAL BATTERIES use the lithium metal as one of their electrodes.
- Whereas LITHIUM-ION BATTERIES store their lithium ions in a different electrode material.
Lithium ions are very small and light, and so they can move easily through batteries. This helps lithium batteries store large amounts of energy, but in relatively small and lightweight packages.
More Information