Battery degradation is the gradual loss of a battery’s ability to store and deliver energy. Over time, all batteries – whether in phones, cars, or backup systems – become less effective.
Understanding battery degradation helps us become more proactive, and even slow it down. But the bottom line is we are talking about a natural process here, although its rate also depends on how we treat the battery.
What’s at the Heart of Battery Degradation?
Chemical changes inside batteries are the core reason why they deteriorate with use. When they are new, these chemical reactions move ions between electrodes to generate power efficiently.
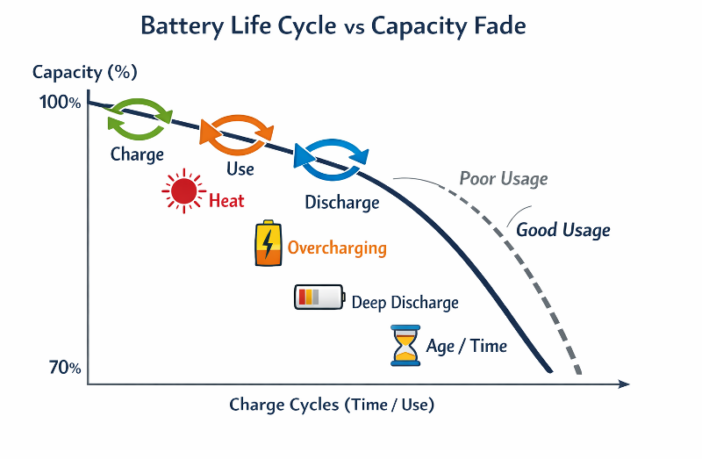
But these reactions become less efficient with repeated battery use. Materials can degrade, form unwanted deposits, and / or lose their structures. Heat, overcharging, deep discharging, and long periods of inactivity can all accelerate this decline.
Understanding Batteries Are Not All The Same
However, different battery types degrade in different ways, for example:
- Lead-acid batteries may suffer from sulfation, where crystals build up and reduce their efficiency.
- Nickel batteries experience ‘memory loss’. They ‘forget” their full capacity if not fully discharged occasionally.
- Lithium-ion batteries, widely used in modern electronics face their own challenges over time.
They deteriorate due to the growth of the solid electrolyte interphase, as well as stress from repeated recharging cycles. High temperatures, and being fully charged for long periods are also bad for them.
Sensible Use Can Slow Degradation
Sensible habits can slow degradation, despite all those issues. Things like avoiding extreme temperatures, not fully draining batteries, and using appropriate chargers can all help extend battery life.
Understanding battery degradation has become increasingly important, as the world relies more on energy storage. Better battery design and smarter usage can combine to make batteries last longer, and reduce waste too.
More Information