Lithium nickel oxide degradation has baffled battery scientists for decades, until now. That’s because this promising cathode material for lithium-ion batteries degrades, and breaks down during repeated discharging. Scientists at University of Texas in Dallas, have discovered why this is happening, and are trialing a solution for the problem.
Why Lithium Nickel Oxide Batteries Break Down
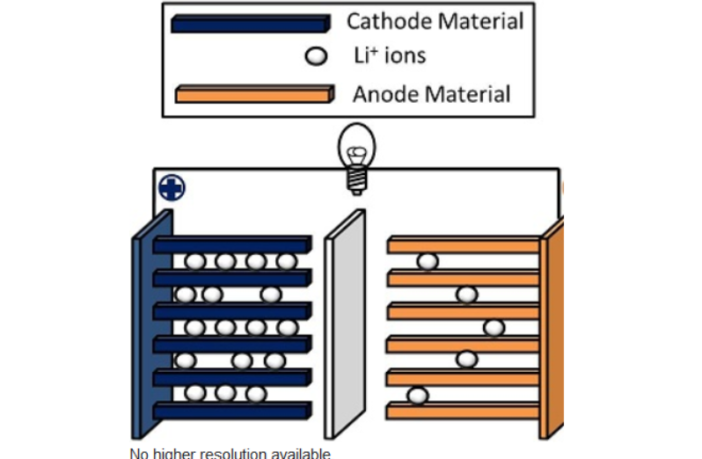
Electrical current in lithium-ion batteries, flows from the positive cathode to the negative anode. This anode, which is typically carbon, holds lithium at a higher potential of the two electrodes.
The positive cathode is typically a more complex compound of materials, including scarce, expensive cobalt. The University of Texas scientists hope to replace this cobalt with lithium nickel oxide.
The lithium-ions return to the cathode through the electrolyte during discharging. An electrochemical reaction simultaneously sends electricity through an external circuit during this process.
This chemical reaction causes the lithium nickel oxide cathode to become unstable, crack, and trigger the degradation. However, the University of Texas scientists have found a way to reinforce the cathode.
Moving Towards Longer-Lasting L-Ion Cathodes
The scientists narrowed the cause of the degradation down to a chemical reaction, involving oxygen atoms in the lithium nickel oxide. “Now that we have a clear understanding of why this happens, we’re working on a solution.” says Kyeongjae Cho, professor of materials science and engineering .
His team developed a theoretical model to reinforce the cathode, by adding a positively-charged ion. This should alter the material’s properties by creating a reinforcing ‘pillar’. If they get this right in practice, then our batteries in phones and electric cars could last longer.
The team’s next step is to manufacture a small amount of modified cathode material in their laboratory. “Then, we will scale up the material synthesis, and manufacture hundreds of batteries per week” a team member promises. ”These are stepping stones to commercialization.”
More Information
Anodes and Cathodes and Their Roles
Stabilizing High-Nickel Cathodes For L-Ion