Electrode materials must bind firmly together for efficient performance. This has not proven that easy given environmental constraints, and this is holding back progress. A team of scientists from Korea Institute of Energy Research, suggests using a paraffin binder for electrode interfaces in lithium-ion batteries.
The Need for More Efficient Electrode Interfaces
High-performing lithium-ion batteries are moving the frontiers of electrode manufacturing forward. The industry needs more efficient solutions in tandem with more sustainable processes.
Wet methods have been mainstream but interest is faltering. This is on account of carbon emissions and capital outlays. Perhaps this is where a paraffin binder for electrode interfaces could present a viable alternative.
However, paraffin wax – which is the main constituent of candles – is not an obvious first choice. This is because the soft, colorless, solid material derives from petroleum, coal, or oil shale, which we need to move on from.
None the less we do need a breakthrough to more affordable and sustainable lithium-ion batteries. In terms of this approach, a paraffin binder could be a path to building high loading, thick electrodes.
More Advantages of Using Paraffin Binders
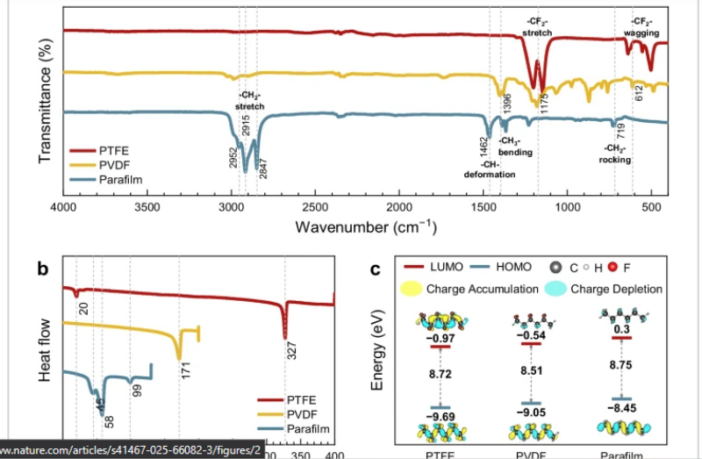
Dry-manufacturing battery electrodes currently depends on using highly-stable polytetrafluoroethylene (PTFE) binders. However, these materials link to high carbon emissions that are leading to global warming. In fact, there is a movement away from PTFE materials.
This approach triggered interest in paraffin-based binders at Korea Institute of Energy Research. Scientists there focused their attention on a dry-mixed Parafilm® M product, that laboratories already use as a sealing film.
This option presented the following advantages:
- Reduced cost and impact on emissions and global warming.
- Chemical stability owing to robust molecular structure.
- Processing at around 60 ºC / 140 ºF, reducing energy profile.
Parafilm® M proved to be an effective primary binder in the Korea Institute of Energy Research laboratory. It simplified battery electrode manufacturing, and contributed to a high capacity product.
Moreover, the resulting electrode also operated for over 1,000 discharge-recharge cycles. Taken together, these results suggest that paraffin binders for electrode interfaces are worth exploring further.
More Information
Hidden Binders in Lithium-Ion Batteries
Polymer Electrode for Water-Based Batteries
Preview Image: Chemical and Thermal Properties
Research Report in Nature.Com Portal
Commentary on Tech Explore Website
.