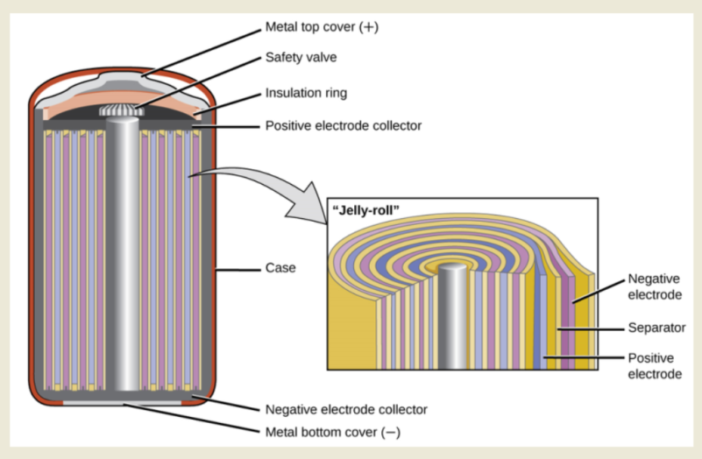
A battery is an electro-chemical reaction between different materials, occurring inside a case. We can tap into this energy via its electrodes, and this power becomes electricity as we receive it. However, a battery does not have unlimited capacity to keep delivering, unless we can reinstate it to its original condition – in other words recharge it. We share battery inner secrets here, that make all this possible.
Subatomic Particles Reveal More Battery Inner Secrets
Electrical energy materializes during a flow of electrons inside a battery. These are subatomic particles of atoms, with negative electrical charges. The chemical reaction inside a battery creates these sub-microscopic items. But they do need to connect to something else to complete the circuit. We need to share more battery inner secrets to describe how the chemistry controls this process.
The two electrodes in a battery are the source and destination for this event. The electrons are there from the beginning, sitting on the negative anode electrode. But they gradually flow across to the positive cathode electrode which attracts them. Recharging a battery reverses this process, as the battery progressively discharges.
However, there is one more important element missing in our description. The negative electrons have a natural inclination to move to the cathode, but they only do so very gradually. Therefore, we need a third element for this to happen in electrically-meaningful quantities. This third element is outside the battery!
How Events Outside a Battery Energize It
We control the electron flow when we connect the battery terminals to an external circuit. Of course, that circuit must provide adequate resistance. If its impedance is too high, then the battery remains dormant. While if it is too low, a spark could tell us we caused a short circuit. As Australian Academy of Science puts is, it must provide a matched, easy pathway.
You may already know the last of our battery inner secrets. When we recharge a rechargeable battery – not all batteries are – we send the electrons back to the anode. Although the battery gradually ages in the process, until it has used up all of its electrons.
More Information
Battery Fundamentals and Flow of Electrons