Batteries are made up of three parts: cathode, anode, and electrolyte. Electrolytes serve as a channel to conduct electricity; it allows ions to travel between the cathode and anode. Composition of electrolytes depends on the type of battery: a lead-acid battery is composed of 35% sulphuric acid and 65% water, alkaline batteries have alkaline solution, and lithium batteries use special organic electrolyte.
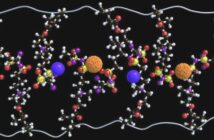
Newly conducted research at the Department of Energy’s Oak Ridge National Laboratory formulated a new double-duty electrolyte. According to Chengdu Liang, one of the authors of this study, “this bi-functional electrolyte revolutionizes the concept of conventional batteries and opens a new avenue for the design of batteries with unprecedented energy density.”
The newly formulated electrolyte has two functions: ion conductor and a cathode supplement.
Because of this dual function, it produces batteries that are more stable and last longer. When the battery is fused with a solid lithium thiophospate electrolyte it produces a higher capacity. A higher capacity means adding years to the battery’s lifespan.
This development is very useful in devices that require a long lasting battery such as cardiac pacemakers, radio-frequency identification devices, remote systems, and sensors.
This research has been published by Ezhiylmurugan Rangasamy, Juchuan Li, Gayatri Sahu, Nancy Dudney and Chengdu Liang in the Journal of the American Chemical Society with a title “Pushing the Theoretical Limit of Li-CFx Batteries: A Tale of Bi-functional Electrolyte.”
Related Article