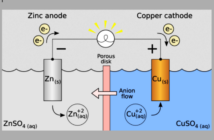
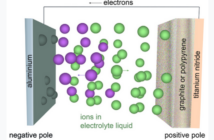
A live battery with a positive charge has active materials containing chemical energy. But this energy is not the same as electricity yet. We must first connect the battery terminals to an external circuit so electricity can flow. We get back to battery basics in this series, to help us understand the direction electro-chemical battery research is trending.
More Battery Basics: Are Batteries the Same As Cells?
Biology describes a cell as the smallest structural and functional unit of an organism. Battery science also calls a single battery a cell, because it is the smallest electrochemical storage unit.
We can connect electrochemical cells in series, or in parallel, to increase their collective voltage, capacity, or both. Benjamin Franklin first called this arrangement a battery in 1749. This may have reminded him of an artillery defence when he gazed down on it.
Our deep-cycle, lead-acid batteries contain a number of cells in series, each delivering a nominal 2 volts of electricity. When battery manufacturers call a single cell a battery, they are not being strictly correct. Although if you asked for a cell they might refer you to the country jail!
The Basic Building Blocks of Every Battery Cell
Getting back to battery basics again, every cell, or cell of a battery has three components without which it cannot fulfill its role. These components are briefly:
- A negatively-charged anode that releases the active electrons to the external circuit.
- A positively-charged cathode that receives the active electrons from the external circuit.
- Electrolyte allowing compensating negative anions to flow in the opposite direction in the battery
Our diagram at the top of this post shows these three essential elements in action. Please come back soon for our next post, as we get back to battery basics in more detail.
More Information
Electrode Chemistry Inside a Storage Battery