Niobium is a transition metal, with high electrical conductivity at room temperature. This makes it a premier element for superconductivity. The metal also plays a minor role in lithium batteries, but has been too unstable to do more. Researchers challenged this assumption, and watched niobium batteries win through before their eyes!
How Did Niobium Batteries Break Through?
Researchers at Sao Paulo University in Brazil, found a way to ‘tame’ niobium in a battery. In headline terms, they created a stable environment, in which the metal could react and convert chemical energy into electricity.
The main challenge to date, according to the research coordinator, has been the extremely high reactivity of niobium. For decades, he continues, its ability to oxidize uncontrollably, and block electron transfer seemed an insurmountable chemical barrier.
“Controlling the chemical environment around niobium allows it to operate predictably, reversibly, and stably,” the coordinator explains. “This enables niobium to take a lead role in a battery, that classical electrochemical systems could never do”.
Now we know when niobium batteries broke through, the question is what exactly happened? Niobium metal is abundant in Brazil. It is also capable of exchanging five electrons. This represents a vary high energy potential.
Prior Experience Behind Niobium Breakthrough
A member of the research team previously worked on two projects, that provided these clues to the solution:
- Certain highly reactive metals do not degrade when a particular protein is present.
- Electrochemical reversibility depends on the surrounding molecular environment.
When the team applied this knowledge to niobium, they realized that the metal was not the problem. Niobium batteries won through the moment they knew the problem was the niobium environment.
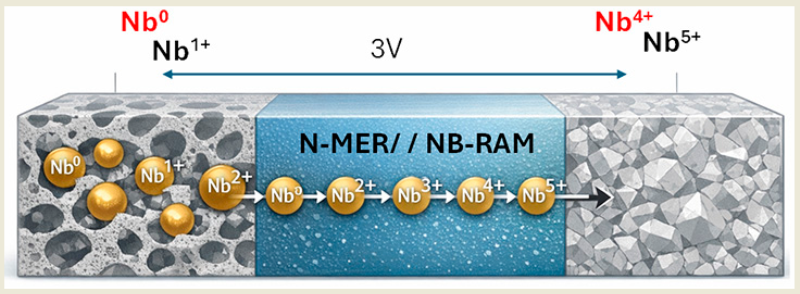
“Rather than trying to ‘tame’ niobium by force,” the coordinator explains, “we created an artificial micro environment inspired by biology. This environment is capable of cooperating with the metal, stabilizing its oxidation states and enabling its reversible operation”.
More Information
How Do Electro-Chemical Batteries Work
Electro-Chemical Reactions Unpacked For You



