Aqueous batteries use a water-based solution for their electrolyte. This makes them safer, more reliable, and less expensive than their lithium-ion counterparts. However, their low energy density, and limited number of charge-discharge cycles they can tolerate, have tended to hold aqueous batteries back. A team from Beijing University School of Advanced Materials, has discovered something most interesting about hydrogen bonds in aqueous batteries.
Hydrogen Bonds In Aqueous Batteries Control Protons
- Ions are whole molecules, or atoms, that have lost or gained electrons. This leaves them with a negative or positive electrical charge.
Metal ions, including lithium, nickel, and zinc, travel through aqueous electrolytes in a diffused, scattered fashion. Thus, the only control the electrolytes exercise is to admit the ions. They have no further control over the speed and direction the ions travel.
- Protons are stable subatomic particles with a positive electrical charge. They are the electrical force which binds atomic electrons together.
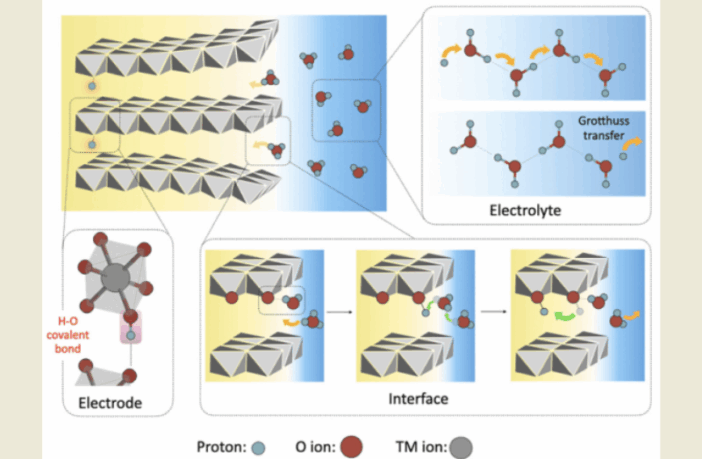
Prof Feng and his team at Beijing University discovered that hydrogen molecules in water (H in H2O) in aqueous electrolytes, exercise greater control over protons passing through them. These hop between hydrogen bonds, enabling faster, diffusion-free travel through aqueous electrolyte.
This discovery positions protons as ideal charge carriers for high-performance aqueous batteries, instead of ions, in future.
Long Term Implications of Hydrogen Bond Discovery
Prof Feng and his team make several recommendations, following their discovery of the role of hydrogen bonds in aqueous batteries. In the first instance, they suggest inserting water-carrying networks of hydrogen bonds in solid-state electrolyte materials.
In the second instance, they air an intriguing possibility in their research findings that we link to below. How about, they ask, we adjust the proportion of acids and negatively charged ions in the electrolyte? It would be interesting to know the effect on stability and proton conductivity.
And finally, Prof Feng and his team suggest modifying the electrode surface, to create ‘proton-bridging channels’. This could improve reaction kinetics, and significantly lower charge-transfer resistance at interfacial points. This research thus appears to open doors for next-gen, proton-based aqueous batteries.
More Information
Topping Up The Ions in Lithium-Ion Batteries
Proton Exchange Technology Breakthrough
Preview Image: Hydrogen Protons Form Stabler Configurations