Did you ever wonder what happens inside lithium-ion batteries? We popped by U.S. Department of Energy and asked them for a straightforward explanation. One without fluff we said. Simply the facts behind the key phrases and concepts we know well. Discover what happens when you use or charge your smartphone here.
Lithium-Ion Batteries and What Happens Inside
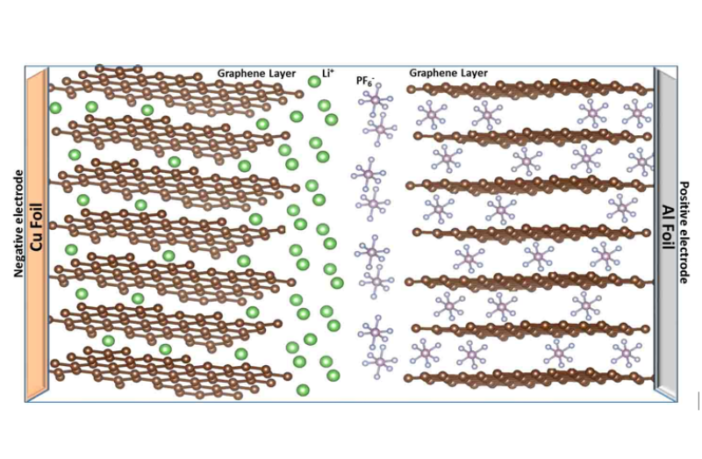
Lithium-ion batteries – and most other electrochemical types – receive, store and deliver potential electrical energy. This is a natural cycle. Trees follow a related procedure when they store energy in the form of carbon. While high up in the sky the sun beams down solar power it captured at the time of creation, and we can recapture and store in batteries.
A related process happens inside lithium ones. They convert the electricity with which we charge them into chemical potential. And then they store this until and application calls for it. All batteries have two separate terminals, anode and cathode, plus a ‘gate keeper’ we call electrolyte that regulates the process.
How Electrochemical Batteries Shuttle Energy
Charged batteries connect to external devices, to complete a circuit between the terminals and allow energy to flow.
- Electrons flow through the external device delivering the electricity it needs to operate.
- While balancing electrically charged ions travel through the electrolyte between the terminals.
- The battery stops working because is ‘flat’, when most of those electrons and ions are consumed.
However, rechargeable batteries – including lithium-ion ones – have a ‘trick up their sleeves’. That’s because their chemistry allows them to reinstate themselves with help from a battery charger. A boost of suitable external electrical energy sends the electrons and ions back to where they belong. The battery is now recharged and ready to run again.
More Information:
Battery Architecture We May Never See
Distributed Energy Resources and Batteries