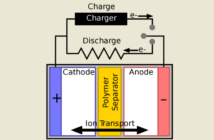
There are various options for cathodes for lithium batteries. In this post we discuss the lithium-ion lithium-cobalt-oxide battery, and debate its usefulness in the bigger picture. Lithium cobalt oxide (LiCoO2) is in quite common use. Here is a useful link if you would like to know more about the structure of the compound scientists also call lithium cobalite.

Some people believe the minor metal we call cobalt is the metal of the future. We already find it in medical isotopes, jewelry, and cosmetics. Some lithium-ion batteries are as much as 60% cobalt.
The Tesla Motors ‘giga factory’ plans to produce enough lithium-cobalt-ion batteries to power 500,000 electric cars by 2020.
This will strain existing cobalt resources. Rechargeable batteries in hand-held electronics already account for 42% of production.
Lithium Cobalt Oxide Battery Stability
Batteries manufactured with LiCoO2 cathodes generally have unusually stable capacities. However these, and the power produced are less than batteries with nickel-cobalt-aluminum (NCA) oxide cathodes. Moreover, they are still prone to thermal runaways resulting from high temperature operation and overcharging.

This weakness seized the public’s attention when a Boeing Dreamliner lithium-cobalt-oxide battery caught fire. This resulted in an emergency landing on January 15, 2013.
This may have had less to do with the material itself, and more to do with battery design and internal circuitry.
In the case of nickel-cobalt-aluminum cathodes, Tesla has found a way to disconnect individual cells with rogue readings. This may be the way forward for Lithium Cobalt Oxide too.
The Way Ahead for Lithium Cobalt Oxide Technology
The Cobalt Development Institute reports that others are following the Tesla example of using lithium cobalt oxide batteries regardless of the minor setbacks. China is pushing already ahead with electrification of transport to counter smog. Samsung is starting to supply the technology for BMW cars and hybrids too.
Related