You may be surprised to hear that nickel cadmium batteries may overheat. They are one of the oldest chemistries with a solid reputation. None the less, overheating can occur, as happened recently on a vessel that was fortunately in port at the time.
A Quick Recap of Nickel Cadmium Batteries
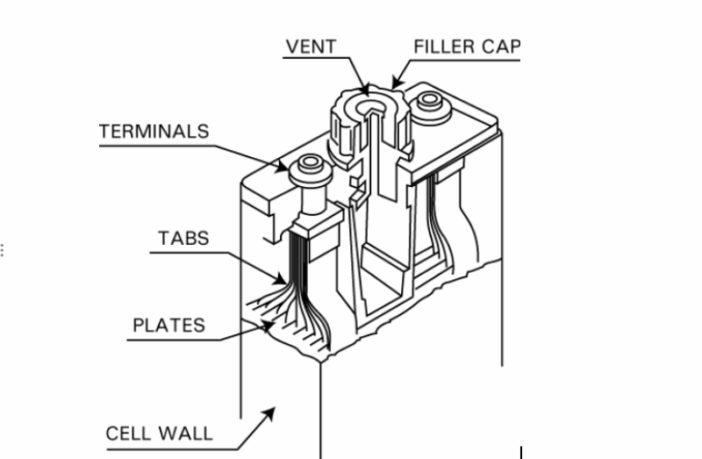
The nickel cadmium batteries we often refer to as ‘nicads’, have cadmium anodes, nickel oxyhydroxide cathodes, and potassium hydroxide electrolytes. This combination delivers relatively high energy density, long cycle life, wide operating temperature ranges, and fast charging.
The cadmium oxidizes to cadmium hydroxide during discharging, as the nickel oxyhydroxide becomes nickel hydroxide. We can reinstate the original composition by recharging the battery,
How Nicad Batteries May Occasionally Overheat
Nickel cadmium batteries may occasionally overheat in one of several ways we describe below:
- If we overcharge a nicad battery, the surplus current may decompose some water in the electrolyte into hydrogen and oxygen gases. This could increase internal pressure, leading to venting and permanently reducing capacity.
- Rapidly charging a nickel cadmium battery, or discharging it with a heavy load, may increase resistance leading to over heating. If we continue with such high current use without cooling, then the internal temperature could rise to a dangerous level.
- Multi-cell nicad battery packs need to all be in a similar state of charge. A weak or over-charged cell can respond with localized heating. Internal and external short circuits can also occasionally generate heat leading to thermal runaway.
Overheated nickel cadmium cells run the risk of cell rupture and gas release. Although the results are more likely to include reduced performance. Several of the causes we mentioned point to incorrect charging, and overloading nicad batteries.
More Information
Lead-Acid Versus Nickel-Cadmium Batteries