Michigan’s Gov. Gretchen Whitmer confirmed manufacture of Pfizer’s oral treatment will commence in Great Lake State. A $120 million investment will follow the announcement in Kalamazoo on June 6, 2022. This will create 250 new jobs and also expand production of active pharmaceutical materials. The PAXLOVID product is a prescription drug for treatment of mild-to-moderate COVID-19 infections.
Applications for Pfizer’s Oral Treatment in United States
U.S. Centers for Disease Control and Prevention (CDC) issued a health advisory on May 24, 2022 (see link below). We are not medical experts, and we therefore provide what follows for information only. In high-level summary terms CDC advises:
- PAXLOVID is suitable for early-stage treatment of mild-to-moderate COVID-19 cases, and helps prevent hospitalization and death.
- However, there have been instances of mild COVID rebound independent of the treatment and vaccination status.
- But transmission may be managed during infection by following CDC’s guidance on isolation, and other precautions such as masking.
Therefore PAXLOVID is not a vaccine or other preventative measure, and it could thus be unwise to drop all precautions simply because ‘there is a fix when we need it’.
How Does PAXLOVID Work in Practice?
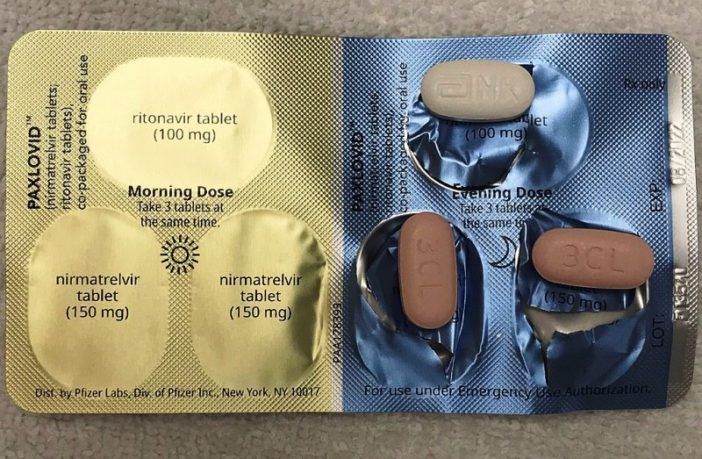
PAXLOVID is Pfizer’s trade name for the combo drug Nirmatrelvir / Ritonavir. It is a dual oral treatment taken by mouth containing these two active ingredients. Moreover U.S. Food and Drug Administration (FDA) authorized its use for COVID-19 treatment in December 2021.
However, FDA does not permit its use for helping avoid infections, or for treatment of patients with severe / critical symptoms. But United Kingdom also approved PAXLOVID in December 2021, and Canada in January 2022.
The drug, which is available in tablet form is furthermore only suitable for people twelve years and older, and weighing at least 88 pounds. But it is not appropriate for women who are pregnant, or who could become pregnant because they are not using contraception according to Wikipedia.
Breaking News
Europe Faces a Higher Risk of Measles Cases
Could COVID Conversion Messages Convince
Preview Image: Combo Package of PAXLOVID
U.S. CDC PAXLOVID Advisory Notice
Pfizer Press Release June 6, 2022