Johann Christian Poggendorff was a German physicist born in 1796, and with a particular interest in electricity and magnetism. He originally trained as an apothecary, meaning he provided medicines to physicians and surgeons. However, his orderly mind soon turned in the direction of the new discipline, Science. Along his life’s journey he invented the Poggendorff Cell in 1842.
The Poggendorff Cell Was an Intriguing Distraction
Johann Poggendorff’s invention turned out to be a side-shoot to battery history, which bloomed briefly, withered, and died. Nonetheless, it contained elements of brilliance that others explored later. Among these inspirations were separating the electrolyte from the depolarizer, thereby eliminating one of the weaknesses of Monsieur Callaud’s Gravity Cell.
A Brief Introduction to Johann Poggendorff’s Apparatus
Poggendorff used dilute sulfuric acid as electrolyte, and chromic acid as the depolarizer to prevent gas collecting at the electrodes. However, he first mixed these together to create a new electrolyte, thereby eliminating the need for a porous pot. And then he turned his attention next to the electrodes at the heart of every battery.
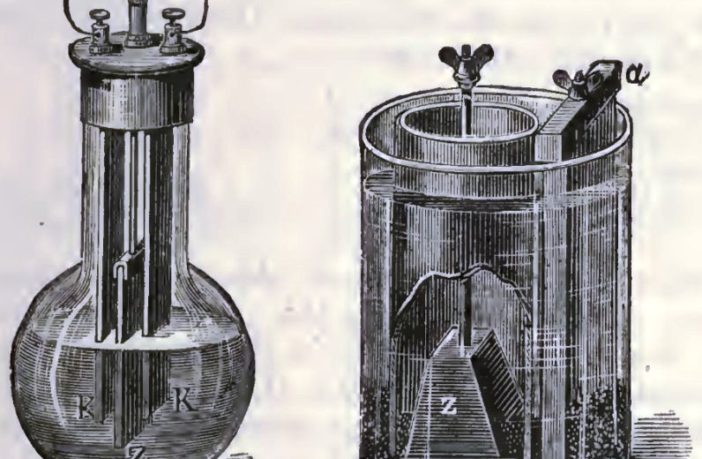
- Two carbon plates in parallel formed the positive cathode electrode.
- While the negative anode was a single zinc plate inserted between them.
- This assembly was in a jar containing the new electrolyte solution.
- A mechanism enabled the operator to raise and lower the zinc anode.
This arrangement helped restrict the natural tendency of the acid to react with the zinc.

Applications for Poggendorff’s Ingenious Invention
The Poggendorff Cell delivered 1.9 volts, making it popular with researchers for many years. And this was particularly because they valued its higher output, consistency, and lack of annoying fumes. However, two distinct disadvantages remained:
- First, the glass container was fragile and required careful handling.
- And secondly, researchers disliked having to raise and lower the zinc plate
During its popular heyday the cell acquired the names bichromate cell, and less commonly, chromic acid cell. Our image shows two later variations, because we could not find a drawing of Poggendorff’s original invention.
More Information
A Porous Pot Cell in a Copper Can