The birth of the button cell battery did not come when alkaline batteries arrived. In fact, it happened in 1942, when Samuel Ruben developed a mercury button cell for use in military metal detectors, munitions, and walkie-talkie radios.
Ruben was just 32-years-old at the time. He cut his teeth on ham radios, but went on to register over 200 patents and established the Duracell company. Although he had no formal qualifications, having dropped out because he could not cope with the stress.
The Button Battery Lived on After the War
Ruben’s mercury button cell was a resounding success, because it delivered a steady output throughout its ten-year life. When the war was over, his military button battery found a second purpose in small electronic devices,like cardiac pacemakers, wrist watches, and hearing aids.
However, the shape of mercury batteries diversified as new applications evolved. So at one time there were also rectangular mercury batteries for transistor radios, and mercury multi-cell packs for industrial applications.
How The Chemistry Worked in Mercury Batteries
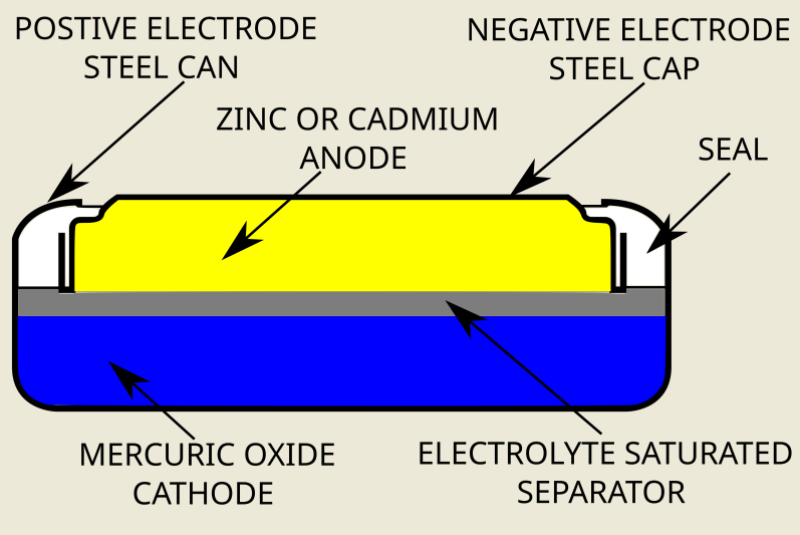
The birth of the button cell battery came about after Samuel Ruben developed his novel chemistry:
- The cathode was either pure mercury oxide, or mercury oxide blended with manganese dioxide.
- Mercury oxide is not a conductor, and so Ruben added graphite to the mix to complete the cathode.
- The anode was pure zinc, separated from the cathode with porous material or paper soaked in the electrolyte.
- The electrolyte was sodium hydroxide or potassium hydroxide, providing two options.
- Sodium hydroxide electrolyte gave nearly constant voltage at low discharge currents.
- While potassium hydroxide electrolyte delivered constant voltage at higher currents.
Days of Mercury Button Cell Battery End
The birth of the button cell battery gave fresh impetus to the development of portable energy devices. However, the mercury in mercury batteries was always toxic, and this would eventually catch up with them.
The 1991 European Commission directive 91/157, prohibited the marketing of certain types of batteries containing more than a certain amount of mercury. The United States followed suit the next year.
The days of mercury oxide batteries were numbered. However, Samuel Ruben’s button cell design lives on. Button batteries using zinc-air, silver-oxide, lithium, and alkaline chemistries have quietly and persistently taken mercury’s place.
More Information
Safe Battery Management and Disposal




