The traditional method for testing for viruses during an epidemic is processing one sample at a time. That’s because laboratories want to avoid mismatching results and people. However, on July 19, 2020 the Food and Drug Administration (FDA) agreed to allow pooling samples for testing during COVID-19. This was because of an urgent need to complete more tests faster.
Faster Results from Pooling Samples for Testing During COVID-19
Efficient contact tracing depends on speed through all the steps in the process. Laboratory reports can take hours, even days to materialize due to limited laboratory capacity. Pooling samples for testing during COVID-19 relieves the pressure, by allowing laboratory testing in series, not parallel.
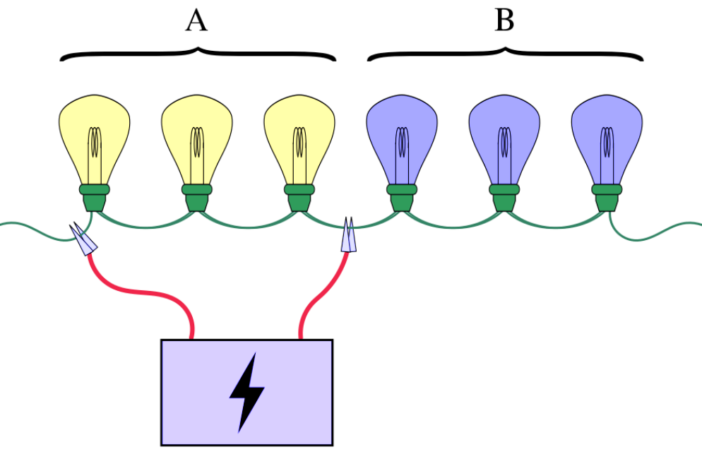
Pooling samples follows the same logic as group testing to find electrical faults. One light bulb in the image at the top of this post is faulty. A split-method group test shows this bulb is in Group B. This reduces the potential number of tests required from five down to a maximum three.
How Sample Pooling Speeds up COVID-19 Testing for Faster Results
The Food and Drug Administration agreed to pool COVID-19 testing to increase processing speed in a similar way. Moreover, it agreed laboratories could pool up to five asymptomatic samples in parallel at the same time. If the pooled result was negative, this would confirm none of the five people had the infection.
However, if the result was positive, then the laboratory would have to re-test all five of the samples individually. The current rate of tests compared to infections indicates pooling samples for testing during COVID-19 will save time and money.
The Food and Drug Administration especially recommends this technique for areas where the known disease prevalence is low. This method may help re-open schools and businesses faster, and get America back on track sooner.
Related
Diagnostic Test for People Without Symptoms
Six Different Clusters of COVID-19 Symptoms
Preview Image: Group Testing Light Bulbs