The term ‘traffic jam’ may have originated on a cattle ranch in 1883, when cowboys were directing a herd through a narrow gate. Whether this is true or false is a matter for debate. However, nowadays we use the term to describe slow moving traffic that stops and starts. Preventing a lithium traffic jam smooths out the flow of ions.
Rachel Berkowitz is a corresponding editor for Physics Magazine based in Vancouver, Canada. She posted an article on April 23, 2024 describing how researchers observed lithium ions become trapped in a cathode, reducing battery performance. The team were using in-situ x-ray at the time, and saw this happening before their eyes.
Degrading Cathodes in Lithium-Ion Batteries
All lithium-ion batteries degrade as their cathodes crack and deteriorate in response to stress. Researchers had hoped to observe this directly during charging and discharging cycles, but this has been tricky until now. This was because batteries are ‘multi-layer structures with buried interfaces’.
Berkowitz describes how researchers devised an x-ray technique, ‘to observe industry-grade versions of nickel-rich lithium-ion batteries in real time’. We investigate this further and describe how this lead to an idea for prolonging battery life.
Preventing Traffic Jams in Lithium-Ion Batteries
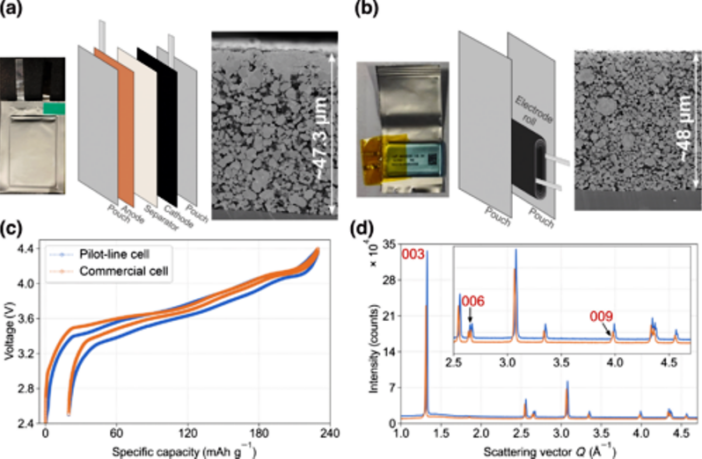
The researchers used purpose-built nickel-rich lithium-ion batteries that they manufactured on a ‘pre-commercial production line’. They then put them through what Berkowitz calls ‘accelerated charging-discharging cycles’.
This process exposed the batteries to the same ‘aggressive voltages, charging speeds, and charging depths’ that commercial batteries experience’. The real-time data they sourced using x-ray diffraction machines disclosed the following:
- Nickel-rich cathodes undergo chemical changes at high voltages.
- The nickel-rich surfaces lose oxygen, while the bulk oxidizes.
- This change causes the buildup of an oxygen-poor surface layer.
- This layer traps lithium ions resulting in a 10% capacity drop after 100 cycles.
‘You end up with a reduced rock-salt nickel oxide crust, about 2 to 5 nanometers thin,’ observes a member of the team. ‘This acts as a kinetic trap slowing lithium extraction and insertion.’
Slowing the recycling rate only went part of the way to preventing a lithium traffic jam. However, doping or coating the cathode surface with aluminum or aluminum oxide ‘might add a protective layer that passivates the nickel surface and prevents lithium traffic jams’.
More Information
Passivation on Negative Battery Electrodes
Battery Fundamentals and Flow of Electrons
Preview Image: Diagram From Research Report