We call a battery a battery, because Benjamin Franklin thought early electrochemical cells standing in a box looked like artillery cannons. Various types of battery chemistries evolved in the nineteenth century, and they needed labels to distinguish them. That brings us around to wondering, why that particular battery name? We discover some early battery types take their titles from real people.
Why Some Batteries Have Particular Names
Did you ever wonder why a particular battery name stuck? Well, here’s your answer, including some early batteries you may have not heard of before:
The Original Voltaic Pile
Alessandro Volta invented the first real battery, the Voltaic Pile, in 1799. This was a simple affair of a pile of copper and zinc disks, with moist material between them. The Pile produced half a volt approximately, which was sufficient to delve further into the phenomenon of electricity.
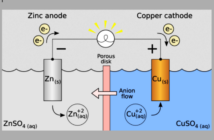
Daniell Porous Pot Cells
John Daniell invented his Daniel Cell battery in 1836. This consisted of a copper pot containing a copper sulfate solution, and a copper electrode. The copper pot stood inside an unglazed earthenware pot, full of sulfuric acid plus a zinc electrode. The output was approximately one volt.
The Grove Telegraph Cell
William Robert Grove developed his Grove Cell in 1839. This had a zinc anode in dilute sulfuric acid, and a platinum cathode in concentrated nitric acid. A porous ceramic pot separated these two elements. Together they produced approximately 1.9 volts ideal for the early American telegraph system.
The Bunsen Primary Cell
Robert Wilhelm Eberhard Bunsen created his Bunsen Cell in 1841. This had a zinc anode in dilute sulfuric acid, and a carbon cathode in nitric or chromic acid, both kept apart by a porous pot. The design produced approximately 1.6 volts, perfect for extracting pure metals, or powering electric arc lights.
The Leclanché Cell Battery
George Leclanché invented the Leclanché Cell in 1866. His original design incorporated a zinc anode, a manganese dioxide cathode, and an ammonium chloride electrolyte in a glass jar. This arrangement delivered approximately one-and-a -half volts for telegraphs, signaling, and electric bells.
These Particular Batteries Did Not Last
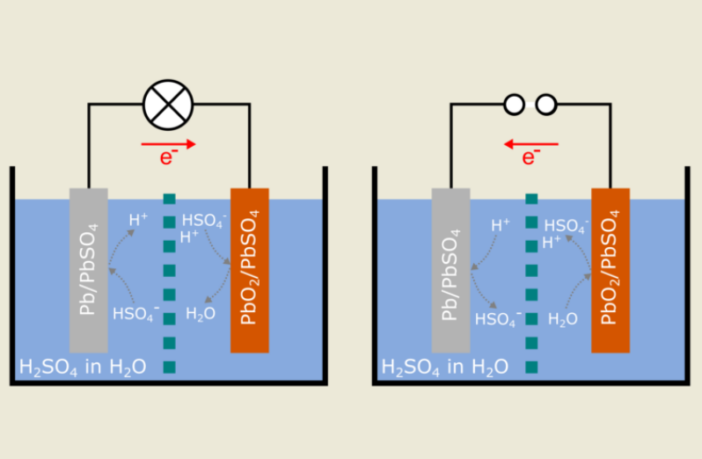
Those early batteries became too cumbersome after utility-generated electricity arrived. They soon fell by the wayside in the face of the rechargeable lead acid chemistry, Gaston Planté came up with in 1859.
Camile Alphonse Faure followed through with a fully portable, stackable two-volt rechargeable lead-acid battery in 1881. That was the year batteries entered main-stream life, and the golden age of batteries began!
More Information
Daniell Cell Solves Hydrogen Bubble Problem