A safer nonflammable battery electrolyte has been marking time at University of Tennessee in Knoxville for two years, while researchers there battle to find a way to finally assemble it. This is a stark reminder of the painstaking processes our scientists follow, as they strive to advance our world of batteries.
Potential of Safer Nonflammable Battery Electrolytes
Many of our modern technologies rely on the efficient transfer of lithium ions between electrodes in rechargeable batteries. However, this technology can become unstable, and pose a significant safety risk.
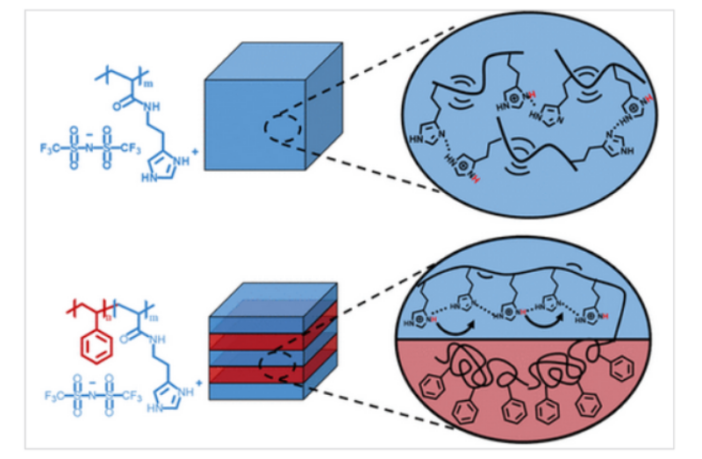
Ion-conductive polymeric ionic liquids (PILs) are a potentially safer nonflammable battery electrolyte, but for one persistent problem. They are by nature soft, and this makes them tricky to assemble and get to work properly.
The team at University of Tennessee in Knoxville trialed several copolymer blends, comprising PILs and rigid polymers. These blocks typically spontaneously self-assemble into ordered nanostructures, that facilitate ion transport and perform well mechanically. But there’s another bridge to cross.
Copolymer Electrolyte Degrees of Conductivity
Battery performance depends on the rate with which electrolytes conduct ions between electrodes. In the case of the block polymers, this rate was significantly slower than that of the soft PIL single-material polymers.
The University of Tennessee in Knoxville scientists, have dedicated the past two years to experimenting with a series of different copolymer electrolytes. They have carefully observed how these self-assemble and conduct ions.
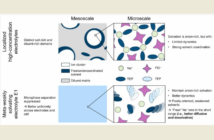
“The self-assembly process has a lot of imperfections,” admits a team member. “We thought it was likely that some of these defects were acting like dead ends, and blocking the movement of ions out of the material.”
The researchers subsequently confirmed that these ‘dead ends’ actually exist, in the form of defects that prevent ion flow. They have published guidelines for controlling this drawback, and developing highly-conductive, robust and safer nonflammable battery electrolytes.
More Information
Biodegradable Battery With Natural Polymers
High Density Polymer Electrolyte Boosts L-Ion
Preview Image: Polymeric Ionic Liquid Block