Solid-state battery electrolytes need careful design. They can’t flow into valleys and cracks in adjacent electrodes as their liquid cousins do. A thin interface interlayer works wonders in situations like this. But the big question is which interlayer material works best in solid lithium-based batteries?
Finding the Right Interface Layer For The Job
A liquid electrolyte naturally contacts an adjacent electrode. Pour it in and the job is done, so to speak. But this is harder to achieve with a mass-produced, solid-state lithium-based battery, where atomic imperfections can creep in.
To complicate matters further, the solid electrolyte and the solid electrode often form new compounds where they make contact. These compounds can get in the way of lithium ions flowing between the two battery materials. This gradually robs the battery of power, and so it fails sooner than liquid electrolyte versions.
Battery scientists have been experimenting with various thin interface layers between these materials, to prevent them touching and reacting. Think of them being like ‘the meat in the sandwich’ if this helps you. In theory, this enables the battery to hold almost full power for longer, meaning improved battery life.
The Thin Material That Works Best With Lithium
Scientists at University of Bayreuth in Germany, formed a joint venture with their colleagues at Hong Kong University of Science and Technology. As you might have expected, their goal was to find out which thin interface interlayer works best with solid-state lithium-based batteries.
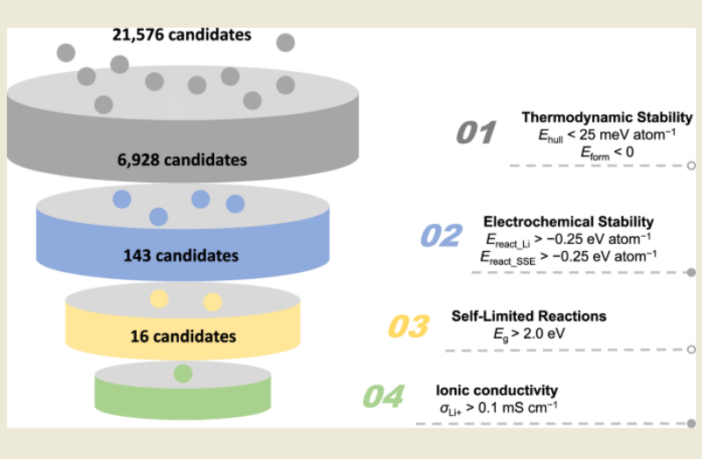
They had literally thousands of options to choose from, thanks to The Materials Project. This is an open-access service that lists material properties to speed up research projects. The team from Hong Kong and Bayreuth spun these options through a high speed computer that delivered their answer quickly.
The optimum solution out of more than 20,000 compounds turned out to be lithium oxychloride. This retained 76% of original battery storage capacity after 1,000 charge-recharge cycles. By comparison, the control solid-state batteries without the special interface layer retained just 5%.
Lithium oxychloride is a compound of lithium, oxygen, and chlorine, already of interest to battery scientists. They are studying the compound as a possible solid electrolyte for lithium-based batteries. In hindsight, its potential as a thin interface interlayer almost seems obvious, just like so many other brainwaves.
More Information
Stable Lithium Anode-Electrolyte Interface
Hidden Interphase in Metal Anodes
Preview Image: Narrowing Down the Candidates