Water is the answer for practically everything, so it makes sense that technology is turning to H2O for more of its technical needs. Researchers are looking at how water can become an electrolyte to power batteries.
The Research:
Researchers from the Swiss Federal Laboratories (Empa’s) Materials for Energy Conversion Laboratory discovered that saline electrolyte should be extremely concentrated to avoid any excess water. For this experiment they used special salt sodium, which is known to dissolve well in water. They combined seven grams of sodium and one gram of water to produce a clear saline solution.
As the water molecules are clustered around the positively charged sodium cations, there are no unbound water molecules left.
Researchers also discovered that the saline solution is sustainable for up to 2.6 volts, which is around twice as much as other water electrolytes. This could indicate inexpensive, safe method of battery cell production.
Potential Problems:
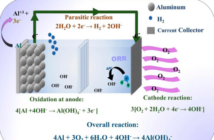
The major drawback with solely water-based solutions is that it is only chemically stable up to a voltage of 1.23 volts. This means that a water cell provides three times less voltage than a standard lithium-ion cell.
Additionally, there is a lack of skilled workers for these jobs, which is limiting its progression.
Trends in Renewable Energy Markets:
Renewable energy sources increase our demand for electric batteries, but lithium reserves are becoming limited. According to the International Renewable Energy Agency, around 9.8 million people work in the renewables sector across the world. This could be because of the manpower needed to generate moving parts, compared to the amount of people needed to facilitate coal or gas plants.
The Benefits:
Electrolytes made from organic materials are prone to bursting in flames. This shouldn’t discount fireproof, or other water-based electrolytes, but most of them are not very reactive. Since this water-based technology contains a high concentration of salt, it creates a solid layer of protection that protects the electrodes from decomposing.
Lithium-Ion is On Top:
For the past several decades, lithium-ion batteries have increased in demand, particularly with the rise of mobile phones and electric vehicles. Its price has increased 240 per cent higher. While their market value is clear, their performance has come under equal spotlight. Draining phones are pointing to alternative technologies.
Skepticism Behind This Technology:
Alkaline batteries use water as a major electrode component, which might enable skepticism because they under-perform in cold weather. There’s also issues of feasibility. Water might not gain leverage in major markets because it’s highly nuanced in the battery technology world. If companies can learn to adapt innovative, sustainable ways of production, potential battery hazards and recalls might become a thing of the past.