On first sight the electrolyte plays a secondary role in a storage battery. However, when we delve into it, we discover nothing could be further from the truth. Today we turn to the fascinating topic of the balancing flow of ions in the electrolyte. We take up the story at the point where electrons start to flow through the external circuit.
The Flow of Ions in the Electrolyte Balances Polarity
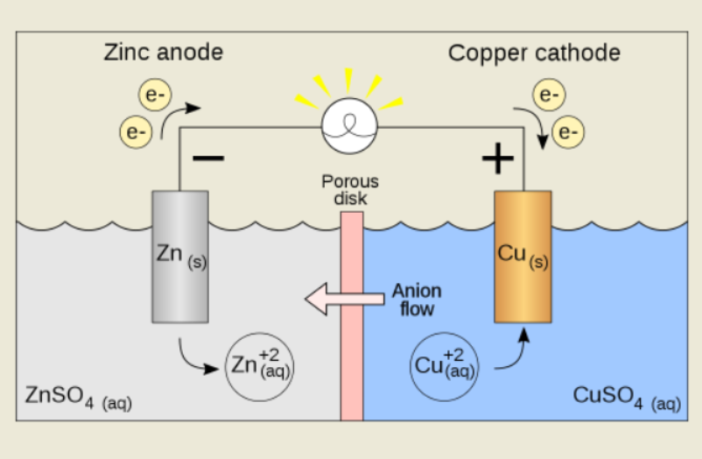
A battery needs a way to balance the negative electrons flowing through the external circuit. It achieves this by releasing a positive stream of ions through the electrolyte inside the case.
- The cathode needs to balance the negative charge of the electrons it receives
- It does so by attracting the positively charged ions emerging from the electrolyte
- Although it could also release the negatively-charged ions into the electrolyte
Therefore, and this gets us closer to the core of the matter, we have two counterbalancing flows in battery chemistry. Negatively charged electrons flow through the external circuit. While positively charged ions flow through the internal electrolyte to restore the polarity, and maintain the overall reaction. This is the magic of battery chemistry in action!
The Limited Life Cycle of a Single Battery Cell
Nothing lasts forever in our imperfect world. For if it did, then the unhindered flow of ions would coat the surfaces of the electrodes, and the reaction could not continue. That’s why most battery cells have a barrier mechanism to moderate the flow. Moreover, when we interrupt the external circuit, the flow of electrons and ions pauses.
But as we use up the battery charge, other chemical products appear. These increase internal resistance that progressively throttle the efficiency of the entire process. The reaction gradually slows until we reach the point where the cathode and the anode can no longer perform their work. The battery gradually discharges to the point where it stops working.
More Information
Build a Battery-Power Electric Train