The earliest electrochemical batteries produced the first electricity humans ever made. Hard to believe? Well this was over two centuries ago, when what we see around us was not even a dream. Nowadays, batteries are mediators between electricity supply and devices. They adjust the input voltage while storing it, and make devices portable. We aim to explore this world of batteries in a short series of posts.
Hidden Secrets in the World of Batteries
There are a host of tiny miracles we call electrons inside battery cases. These allow us to store electricity, and use it to power our portable devices wherever we are. Our world of batteries is expanding rapidly to meet the demand for mass energy storage. Yet all batteries work the same way as penny button cells.
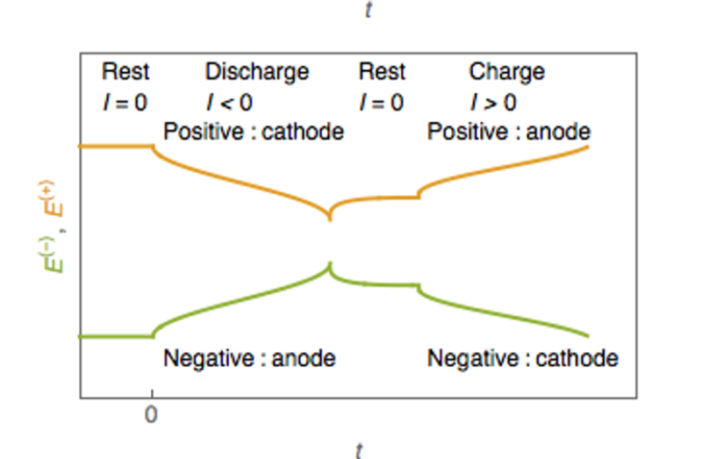
- All batteries have three basic components causing chemical reactions.
- These are a negative anode, a positive cathode, and an electrolyte.
- This electrolyte causes a reaction involving the anode and the cathode.
- This occurs when we connect the negative and positive through a circuit.
- Electrons emerge at the negative terminal, and flow through to the positive.
- Those electrons are the energy we call electricity, that powers our devices.
Primary and Secondary Batteries in Our World
There are limits to the supply of electrons a battery can hold. But we may be able to replenish them, depending on the design of the battery.
Primary, Non-Rechargeable Batteries
Primary batteries are a reminder of our discredited throw-away society. Folk still use them because they are cheap, although you should not find anything like that in our battery factory. Primary batteries stop working after they use up their supply of electrons. But we can send them to the recycling yard and re-use their minerals.
Secondary, Rechargeable Batteries.
We can replenish the electron supply in a spent secondary battery, by recharging it from an external source. Not perfect, but it is a step in the right direction.
Most secondary batteries are either lead-acid, zinc-air, nickel-cadmium, nickel-metal-hydride, lithium-ion, lithium-ion-phosphate, or lithium-ion-polymer. Each plays an essential role in the emerging world of batteries today.
More Information
Vacuum Tube Batteries in Bygone Times