Batteries take the chemical energy in their materials, and change this into electrical energy. We have written thousands of posts on this blog about these wonderful electrochemical devices. We learned a great deal more about batteries and how they work, as we traced breaking news about this evolving technology.
A Return to The Inner Workings of Batteries
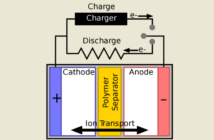
All batteries, including the one perhaps powering your phone as you read this, contain an anode, a cathode, a separator, and an electrolyte that combine to produce electrical energy:
- The anode is the negative terminal that stores the electrochemical energy, and releases electrons in a process we call oxidation.
- The cathode is the positive terminal that accepts these electrons. It receives them in a process we call reduction.
- The separator prevents the cathode and anode from touching directly. Its job is to avoid a short-circuit releasing all the energy at once.
- The electrolyte allows ions to move from the anode to the cathode during discharging, and move back again during recharging.
So much for the basics of all batteries. Please stay with us, and learn more about batteries, and how these four components work together inside them.
How The Four Parts of a Battery Work Together
- Chemical reactions occur at the anode and the cathode when we discharge (use) a battery.
- In the first instance, the anode releases subatomic electron particles to the cathode via an external circuit.
- In the second, ion atoms flow the other way through the electrolyte, to restore the electrochemical balance.
- These two counter-balancing flows generate the electrical current that powers portable devices.
When we recharge a battery (if it allows us) we reverse the ion flow, by passing an electrical current through the battery in the other direction. However, a few ions become trapped every time this happens. Eventually the battery is no longer able to produce sufficient energy to power our electrical device.
More Information
Anode-Free Metal Batteries – Back to Basics