The original design for Planté’s lead battery called for flat plates comprising pure lead sheets. Since then, battery designers discovered battery capacity is proportional to the electrode surface area in the electrolyte. We discuss subsequent steps to increase the capacity of negative and positive lead battery plates. This is quite a complex topic and may spill over into a second blog.
Increasing Capacity of Lead Acid Battery Plates
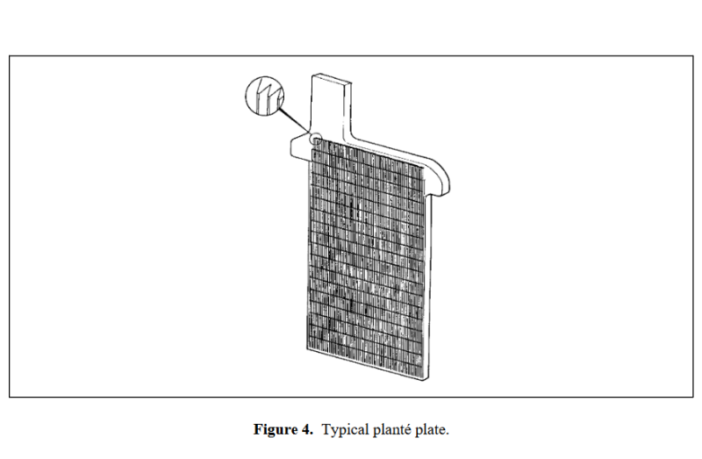
Planté experimented with grooved, and perforated plates to enhance his design. Although this method, as our first image shows had its limits. The most common approach nowadays involves turning the active material into a paste, with the appearance of a sponge full of tiny holes.
Lead acid battery manufacturers apply this paste to a frame or grid structure that mechanically supports it. The electrolyte is then free to enter all the tiny holes in the sponge, thereby increasing the effective capacity of the battery.

The negative and positive lead battery plates conduct the energy during charging and discharging. This pasted plate design is the generally accepted benchmark for lead battery plates. Overall battery capacity is increased by adding additional pairs of plates.
Bolstering Negative and Positive Lead Battery Plates
A pure lead grid structure would not be able to support the above framework vertically. Therefore, battery manufacturers use a lead alloy material for added strength, and enhanced electrical properties. The commonest additives are antimony and calcium, although tin may be added to improve discharging and recharging.
Lead-antimony versions may deep cycle more often than lead calcium ones. Although flooded versions require more frequent maintenance towards their end of life. Lead-calcium versions have lower self-discharge rates and draw less current on float charge. But frequent over-charging can lead to plate buckling and rupturing.
More Information