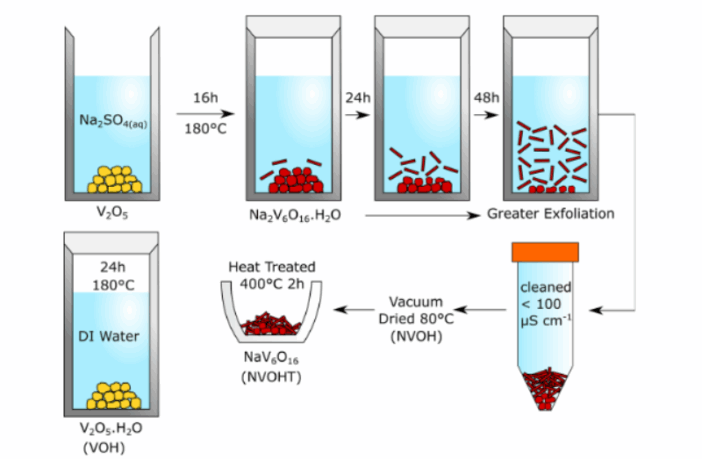
Sodium vanadium oxides make promising cathode material in sodium-ion batteries. These oxides contain water in their original form, but chemists remove it to enhance structural stability and increase conductivity. However, recent research at University of Surrey in England suggests retaining water in sodium-ion batteries, brings some surprising benefits.
Water in Sodium-Ion Cathodes Improves Performance
Sodium vanadium oxides contain water in their original form, that is nanostructured sodium vanadate hydrate. Battery chemists traditionally remove this moisture using procedures such as vacuum drying, thermal annealing, and controlled dehydration.
Those procedures cost time and money, so there might be a saving if we could avoid them. Perhaps this is why the Surrey researchers investigated the possibility of retaining some of the water. Whatever the case, something remarkable happened when they did:
- There was a major boost in cathode performance in terms of sodium-ion battery capacity.
- The battery also charged much faster, and was still stable after 400 discharge-recharge cycles.
In fact, and this really was the clincher, the ‘wet’ version of sodium vanadium oxide held almost twice as much charge as typical sodium-ion alternatives. This positions the wet version among the best-performing cathodes in the literature.
Completely Unexpected Result of Retaining Water
Lead author and research fellow Daniel Commandeur was ecstatic about the result. “Our results were completely unexpected. Sodium vanadium oxide has been around for years. People usually heat-treat it to remove the water because it’s thought to cause problems.”
He goes on to confirm the result of challenging this tradition. The outcome was far better than they expected. The hydrated sodium vanadium oxide delivered much more powerful performance, and charge stability too. Indeed, the result hints at exciting new uses for sodium-ion batteries.
But there was another, unexpected, ‘side show’ to retaining some water in sodium-ion batteries, when they used seawater in their modified cathode material. The battery removed the sodium-ions from the water during charging, effectively desalinating it.
More Information
Tackling Water Uncertainty in Climate Change
380,000 Cycles With a Seawater Battery