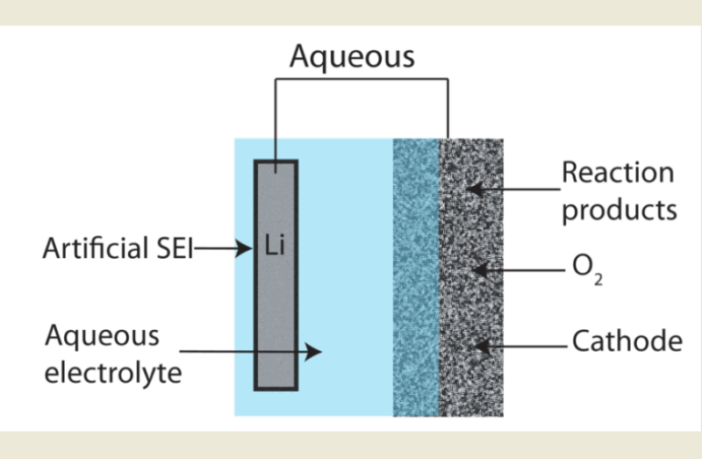
There are two classes of electrolytes in chemical batteries that store electrical energy. The first group of electrolytes are broadly-speaking liquid, while the second are solid. One type of liquid electrolyte comprises a blend of chemicals, while the other is water-based. We discuss the use of sulfate salts in aqueous electrolytes, which are our topic for today.
Free Water Molecules in Aqueous Batteries
Aqueous battery electrolytes should be less expensive, than those that are a blend of chemicals. However, they have a nutritiously short lifespan, which makes them more expensive per unit of electricity over their lifetime. This renders an environmentally-friendly option less commercially attractive.
A team of scientists at King Abdullah University of Science and Technology (KAUST) appears to have resolved this. Perhaps unsurprisingly in hindsight, the problem lies in the water molecules. But first we need to take a brief detour through chemistry:
Three Principles Behind the KAUST Discovery
- Molecules are groups of two or more atoms held together by attractive chemical bonds. A molecules is the smallest unit of a chemical compound that can contribute to a chemical reaction.
- Free water molecules can be ‘rogue’ elements, because they are not strongly bonded with other molecules. They may cause spurious reactions that compromise aqueous battery anodes.
- Battery anodes are one of a pair of electrodes that exchange ions through an electrolyte. An anode is a battery’s contact point with the outside world, while it discharges or recharges.
How Aqueous Electrolytes Benefit From Sulfate Salts
The KAUST team focused their attention on metal battery anodes, which prevented the full return of ions in aqueous batteries. They developed special electrochemical tools, which demonstrated that the root cause was free water molecules in the electrolyte.
The scientists found that adding affordable sulfate salts, such as zinc sulfate, reduced the quantity of free water molecules in aqueous battery electrolytes. And as a result, the ions exchanged more completely between the electrodes.
The KAUST report confirms that the sulfate salts in aqueous electrolytes increased aqueous battery lifespan by more than ten times.”Our findings highlight the importance of water structure in battery chemistry,” a spokesperson observes. “This is a key parameter that has been previously overlooked.”
More Information
Hydrogen Bonds In Aqueous Batteries Exposed
Aqueous Zinc-Sulfur Chemistry Taps Door
Preview Image: Schematic of Aqueous Battery