Batteries evolved down several centuries through the cumulative efforts of many people, most of whose names are lost in time. The origins of lithium battery chemistry followed a similar path with twists and turns. This process began in the 1960’s, when Ford was developing a sodium-sulfur battery prototype for a future electric car.
How Lithium Battery Chemistry Evolved from Sodium-Sulfur
Ford’s ideas were revolutionary. They used liquid sodium and liquid sulfur electrodes, and a solid beta alumina electrolyte. This turned the battery tradition on its head, of solid electrodes and liquid electrolytes.
Construction Physics explains how the ions moved rapidly through the solid beta alumina electrolyte, although Ford did not understand why. Someone else would have to grasp the baton, and take the origins of lithium battery chemistry further.
Fast forward to 1970, when M. Stanley Whittingham was a young chemist working at Stanford University. They asked him to investigate fast ion transport through solid electrolytes, and understand the dynamics.
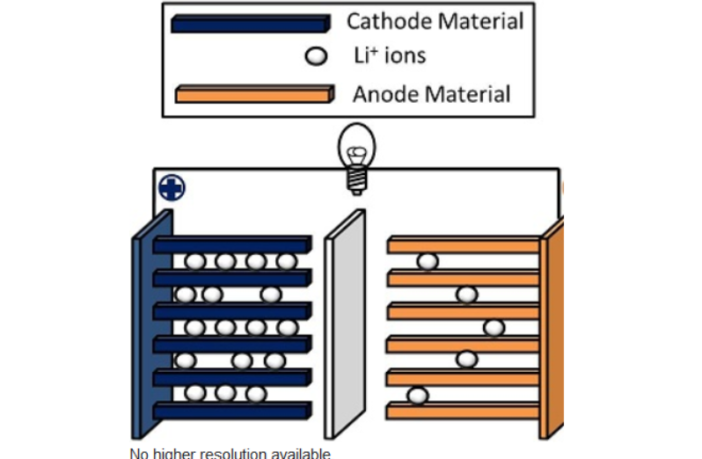
Whittingham built a battery with a solid beta-alumina electrolyte, between two electrodes of tungsten oxide. The ions moved rapidly as he expected. Then he noticed that the ions were intercalating in and out of the tungsten electrode layers for the first time.
The Birth of the Original Lithium-Ion Battery
The Exxon oil company became concerned that oil reserves were topping out in 1972, and began investigating other energy sources. They recruited Whittingham to their team to investigate superconductivity. The young scientist applied his mind to metallic tantalum disulfide, which has superconductive properties.
Whittingham observed high energy reactions when he injected potassium ions into a metallic tantalum disulfide compound. He explored other compounds as potential energy stores, before building a battery with a titanium disulfide cathode, and and a potassium anode.
When the potassium proved hazardous, Whittingham substituted metallic lithium, with lithium salts for the electrolyte. The ions shuttled and intercalated as he expected. But that was only the beginning of lithium battery chemistry.
More Information
How Electrons Travel In and Out of Electrodes