In the previous post, we learned about Gaston Planté’s life as a scientist and his invention of the first lead-acid battery in 1859. In this post, we will discuss further its principle of operation and its impact on the battery technology of today.
Image Source: Magnet Lab Website
What is a Planté Battery?
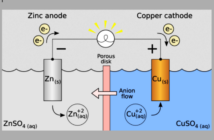
Planté battery is a rechargeable battery and the first of its kind. It is constructed with two electrodes separated by a rubber strip. The negative electrode (or anode) is made of lead while the positive electrode (or cathode) is made of lead oxide. The electrolyte used is a solution of sulfuric acid and water. During discharge, electrons are conducted from the anode to the cathode by the electrolyte. The electric current is then produced powering up the load (an electric bulb, a vehicle, or what-have-you).
This concept of a battery by Gaston Planté was in fact inspired by the Daniell Cell, a primary cell that was only capable of generating 1.1V. Significantly, Planté’s single cell can supply 2V, the chemical reaction is reversible, and a number of cells can be combined in order to produce higher voltage capacity. In addition, his battery only used a single electrolyte for both electrodes compared to the Daniell Cell which used two types. Planté’s concept of the nine-cell battery came a year after his invention of the single cell type. He presented this improvement of the first design to the French Academy of Sciences in 1860.
One disadvantage of Planté battery is that it can only supply voltage for a short period of time. This is attributed to the cathode which has small amount of active lead oxide material available during chemical reaction. This, however, was enhanced by Camille Faure in 1881. Faure used a set of electrodes consisting of lead paste spread thinly on the electrode surface thus resulting in longer chemical reaction.
Planté battery is the foundation of the modern-day lead-acid battery. Today, there are two main categories of 12V (derived from Planté’s nine-cells capable of producing 18V) lead-acid batteries. The first one is the so-called starting battery which supplies short surge of high electric current. The second category is the deep cycle battery which is capable of discharging lower current over a longer period of time.
Related articles: