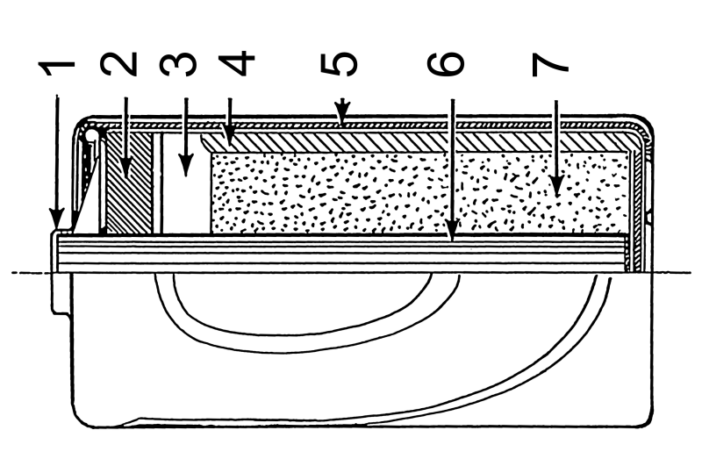
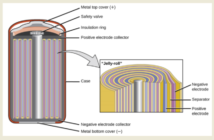
Components of a Dry Cell Battery
A dry cell battery is a single, or multiple electro-chemical cell that converts chemical energy to electrical energy. It contains a ‘dry’, non-liquid electrolyte that may be a paste or other damp medium. A typical structure consists of a zinc metal anode, and a central carbon rod cathode. In this instance the electrolyte is likely to be an ammonium-chloride paste
The Origins of the Zinc-Carbon Non-Aqueous Battery
Carl Gassner was a German physician specializing in eyes and ears, who also liked to tinker with physics and chemistry. Battery-operated doorbells were popular gadgets in 1880, but their cells had short lives.
This was because they were Leclanché-type batteries with liquid electrolyte, that often dried out making them redundant. We’ll never know for sure what caused Gassner to add plaster-of-paris and zinc-chloride to the aqueous solution. One version says a shopkeeper-friend asked him for help, and Gassner’s dry cell battery was the result.
Columbia Dry Cell – First Portable Electricity For The Masses

Dry batteries with single, or multiple cells are a popular choice for small portable devices, even without lithium-ion density. Cost may be the deciding factor, although their non-flammable paste-like electrolyte is also less likely to leak.
Carl Gassner patented his non-aqueous battery in the United States in 1887. However, National Carbide Company (NCC) improved it by replacing the plaster-of-paris stiffener with coiled cardboard. This saved space, simplified manufacture, and created more room for the carbon rod. The company began marketing their ‘Columbia Dry Cell’ in 1896.
The sealed, six-inch, 1.5 volt Columbia Dry Cell was the first ‘battery for the masses’ who could finally afford one. At last, a portable electro-chemical device was available that could store electricity in useful, slip-in-pocket quantities. Our world of batteries had taken a leap forward. and could never be the same again.
More Information
Was Sakizō Yai First With a Dry Cell?