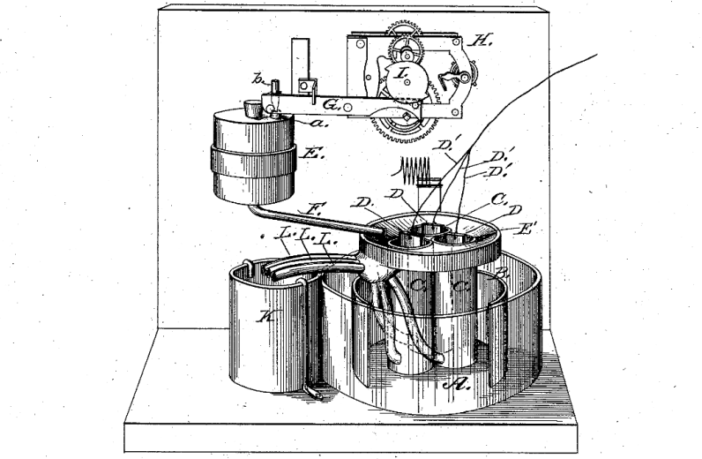
A flow battery comprises two different, dissolved chemical constituents stored in separate tanks. These liquids pump through separate systems sharing a common membrane. Ions pass through this separator, while electricity simultaneously flows through an external circuit. Today we discuss the first flow battery John Doyle patented in February 1880.
What We Know About John Doyle and His Patent
We know very little about John Doyle, whom we believe was a contemporary of Thomas Edison. He filed his patent application US224404 on September 29, 1879. This was for a zinc-bromine version Yuriy Tolmachev writing in Qeios confirms had multiple, refillable cells.
Although this design was quite different from modern flow batteries we know, Yuriy Tolmachev believes recharging would have been possible. However, this would have required additional pumps the patent application does not mention. Therefore we can say with some certainty we agree this was the world’s first flow battery.
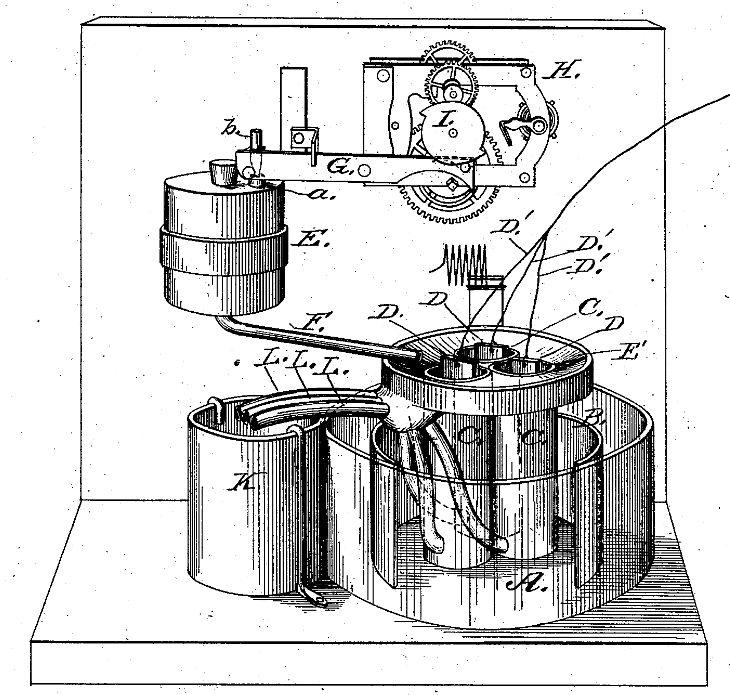
Some Snippets from John Doyle’s Application
IN THE FIRST INSTANCE
- My invention is a series of porous cups grouped together in a single cell of a galvanic battery.
- These contain a series of negative elements connected together to one of the circuit wires.
- A funnel-shaped receptacle receives and conveys the excitant from the reservoir to the said cups.
- A series of overflow pipes conveys the exhausted excitant from the porous cups to storage.
IN THE SECOND INSTANCE
- A galvanic battery powers a mechanism that releases a supply of fresh excitant to the porous cups.
Alas, we have run out of time to get this blog ‘to the printers’. We’ll pause for now. But we’ll be back to describe how this first flow battery worked in more detail in our next post.
More Information
Battery Carbon Footprints Becoming Critical