For two centuries, scientist believed a battery comprises just three components: two electrodes with an electrolyte between them inside a container. However, there is more to them.
Lithium batteries have become commonplace. Yet what happens inside them at a molecular level remains a mystery. That is, until scientists analyzed the SEI layer inside lithium batteries between solid electrodes and liquid electrolytes.
How This SEI Layer Inside Lithium Batteries Is the Interface
Until now, scientists have not really understood what happens at this interface between the liquid electrolyte and solid electrodes. We have assumed that ions pass through it during charging and recharging at molecular level.

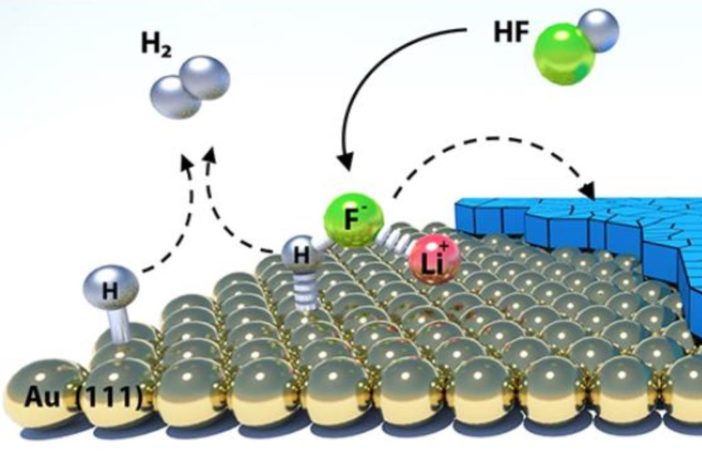
We thought somebody else understood this better, but the truth is nobody did. Now scientists at Argonne Laboratory have released their chemical analysis of the microscopically thin layer inside lithium batteries. This concentration of hydrogen fluoride forms at the interface between the liquid electrolyte and solid electrodes during the first charge of the battery. This may be why we fully charge a new lithium battery fully before we use it for the first time.
More Information About this Solid Electrolyte Interphase
Scientists have known about the SEI layer inside lithium batteries for a while. Now the solid electrolyte interphase is less than a thousandth of a millimeter thin. And nobody previously knew how to monitor it chemically.

Argonne researchers are alive with excitement now that they know how the concentration of hydrogen fluoride works. Their co-principal researcher Dusan Strmcnik told the media, “Battery performance is highly dependent on the quality of the SEI. If the chemistry and the role of individual components of the SEI are understood, the SEI could be tuned to improve battery performance.
“More importantly, such understanding would significantly improve our predictive ability of battery lifetime. This is of extremely high value to an electric car manufacturer,” he added.
Related
Lithium Battery Aging Explained
New Electric Car Battery Decay Caused by Heat
Preview Image: Converting Hydrogen Fluoride Impurity from the Electrolyte into Lithium Fluoride