A team of scientists in Saudi Arabia have discovered something most interesting. Aqueous batteries that use water-based solutions as their electrolytes, have short lives because of loosely-bonded water molecules. The researchers have developed a salt to remove free water from aqueous battery electrolyte, but only in the laboratory.
Where Do These Free Water Molecules Come From?
Free water molecules are units that are not tightly bonded in water-based solutions. This means they can move independently, and trigger harmful reactions at battery anodes.
The researchers discovered how to stabilize water-based electrolytes, and prevent these harmful reactions happening at battery anodes. Their solution extends battery life ten times under experimental conditions.
The Sulfate Salts That Removed the Problem
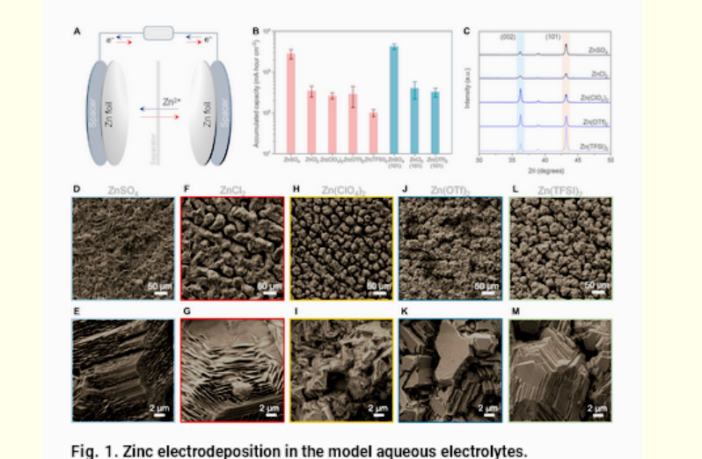
The sulfate salts that removed the free water molecules from the aqueous water-based solutions, are compounds. Chemists form them by combining sulfate anions and organic cations.
The scientists trialed various options first, before settling on zinc sulfate in an experimental zinc-zinc battery. The zinc sulfate acted like a ‘water glue’, they say. It stabilized the water structure and ‘stopped the energy-wasting reactions’.
‘Not only is this solution simple and cost-effective, but early results suggest it may be a universal fix for various types of metal-anode aqueous batteries.’(Science Direct)
Water Structure in Aqueous Battery Chemistry
This discovery reminds us of the important role of water in battery chemistry. Having too many free water molecules frees them to consume battery energy, and damage the anode.
Developing a salt to remove free water, and extend battery life ten times, is a remarkable discovery. It could dramatically improve the prospects of water-based electrolytes, that are non-flammable and healthier for our environment.
“Sulfate salts are cheap, widely available and chemically stable. This makes our solution scientifically and economically viable,” comments Yunpei Zhu conclusively, who conducted the bulk of the experiments.
More Information
Aqueous Zinc-Sulfur Chemistry Taps Door
Hydrogen Bonds In Aqueous Batteries Exposed