We need better solid lithium batteries, if we are to take this promising technology to new heights of reliability. Two key problems with solid lithium batteries still stand four-square in the way of progress.
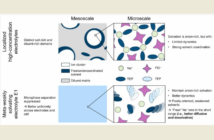
The first of these issues is an inherent weakness in the solid electrolyte, that allows dendrites to take root. These tiny, needle-like formations can cause internal short-circuits between the electrodes.
The second impediment is the inherent weakness in the battery structure itself. There is an imperfect interface between the anode and the electrolyte, leading to poor performance and unreliability.
Researchers Tackle Solid Lithium Battery Issues
On January 9, 2026, the Swiss Federal Office for the Environment announced a breakthrough. Researchers at Paul Scherrer Institute had achieved a practical step towards better solid lithium batteries. This discovery could make them safer, and charge faster, than lithium-ion batteries.
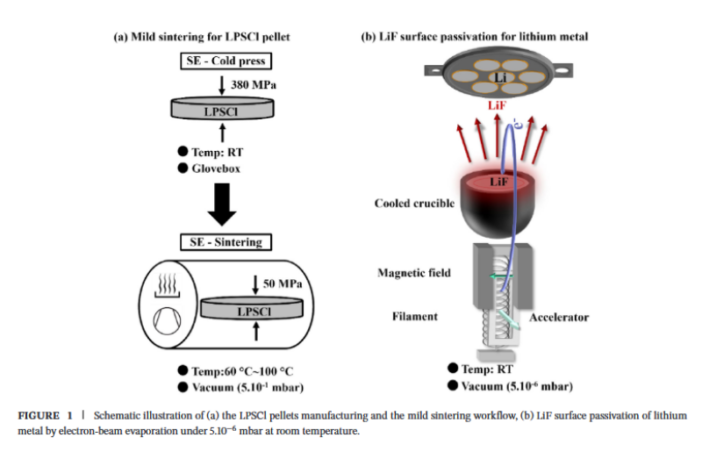
The Scherrer solution involved changing the battery production process in two ways, that together achieved two important advances. The electrolyte became denser and more resistant to dendrite formation. And at the same time the interface between the anode and the electrolyte also improved.
Technical Detail Behind Solid Battery Improvement
What follows is quite technical, although we’ll do our best to unpick it for you:
- The Swiss researchers applied their minds to a particular electrolyte, containing lithium, phosphorous and sulfur.
- This combination allowed fast ion transfer through it, which is critical for high performance and fast battery charging.
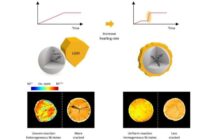
- However, they also needed to make this promising material denser, to eliminate the pores that invited dendrites.
Their solution involved gently compressing the electrolyte material at 80º C / 175º F. This was far lower and gentler that the traditional ‘sintering’ method. The lower temperature and pressure ensured the electrolyte particles formed closer bonds, eliminating cavities inviting dendrites.
The scientists perfected their work by adding a thin, intermediate layer of lithium fluoride between the anode and the solid electrolyte. This further suppressed dendrite formation. Their improved solid lithium battery also achieved 1,500 charge-recharge cycles, with 75% retention of original energy storage capacity.
More Information
Hidden Pathways in Lithium Batteries
Stable Lithium Anode-Electrolyte Interface
Preview Image: Overview of Research Process