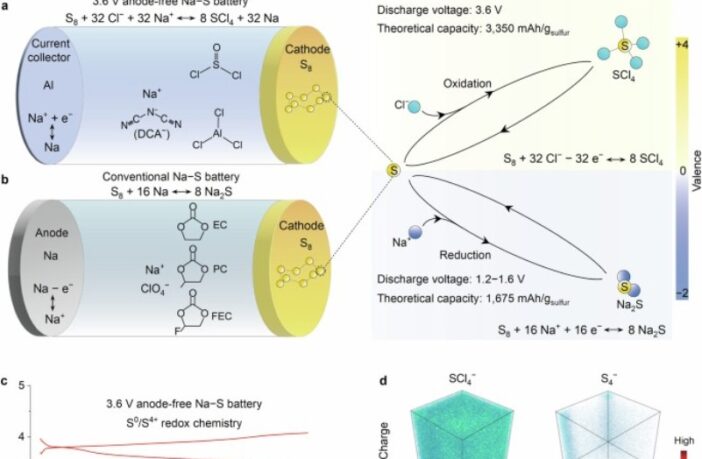
High voltage sodium-sulfur batteries use liquid sodium and liquid sulfur electrolytes. They are relatively inexpensive, and store the same amount of energy per volume as lithium-ion. However, their high operating temperatures limit them to stationary energy storage applications. Researchers in Shanghai, China have just developed a low-temperature version with voltage comparable to lithium-ion.
High Voltage Sodium-Sulfur Breakthrough
Chemistry World remarks how sodium batteries may have crossed a critical threshold, ‘by moving into high-voltage territory’. But is this a practical pathway to low cost, sustainable battery storage? Let’s find out!
Well first of all, both sulfur and sodium are cheap and readily available in large volumes. This makes high voltage sodium-sulfur batteries a potential runner for large-scale commercial storage. This also renders them a worthy partner for environmentally-friendly renewable energy.
But it would be great if we could use this affordable green energy for our watches, phones, and laptops. Unfortunately though, the sulfur sodium chemistry has let us down in that area. You see, low temperature versions produce 1.6 volts at best, and that can’t compete with lithium-ion.
Major Breakthrough For Sulfur and Sodium
The Shanghai researchers developed a novel sulfur chemistry, that delivers a lithium-like 3.6 volts at room temperature. To achieve this, they delved deeply into science that’s beyond the scope of this article. Please follow the links below for the original research on Nature.Com, and Chemistry World’s assessment.
Achieving 3.6 volts at room temperature sounds like a major step forward for affordable, practical high voltage sodium-sulfur batteries. Further innovations achieved in the Shanghai laboratory include:
- Forming a cathode from the super heavy seaborgium element.
- Using aluminum foil as the anode electrode current collector.
- Installing non-flammable chloro-aluminate as the electrolyte.
This arrangement does away with using traditional sodium anodes. Instead, metallic sodium forms temporarily on the current collector during charging. This reduces both weight and cost, while enhancing user safety.
More Information
Breakthrough Battery Anode Material
Simpler Sodium-Sulfur Batteries in Lab